Different types of clinical evidence and study design
The research questionWhat is a research question?
The researcher asks a very specific question and tests a specific hypothesis. Broad questions are usually broken into smaller, testable hypotheses or questions.Often called an objective or aim, though calling it a question tends to help with focusing the hypothesis and thinking about how to find an answer
What makes a poor research question?
a question that matters to nobody, even youhoping one emerges from routine clinical records
the records will be biased and confoundedthey’ll lack information you need to answer your question reliably, because they were collected for another reason
fishing expedition/data dredging – gathering new data
and hoping a question will emergeWhat makes a good question?
Feasible (answerable with a robust method)
Interesting
Novel
Ethical
Relevant
FINER criteria
Real research questionsIs five days’ treatment with
injectable ampicillin plus gentamicinmore effective than
chloramphenicol in children under 5
with very severe pneumonia in low
resource settings?
What is the prevalence of HIV
infection in India, and how manypremature deaths does it cause?
How to focus your question
brief literature search for previous evidencediscuss with colleagues
narrow down the question – time, place, groupwhat answer do you expect to find?
Turning a research question into a proposal
who am I collecting information from?
what kinds of information do I need?
how much information will I need? *
how will I use the information?
how will I minimise chance/bias/confounding?
how will I collect the information ethically?
* sample size – ask a statistician for help
http://www.bmj.com/collections/statsbk/13.dtl
Minimising bias and confounding
Chance - measurements are nearly always subject to randomvariation. Minimise error by ensuring adequate sample size and
using statistical analysis of the play of chance
Bias - caused by systematic variation/error in selecting patients,
measuring outcomes, analysing data – take extra careConfounding - factors that affect the interpretation of outcomes
eg people who carry matches are more likely to develop lungcancer, but smoking is the confounding factor – so measure likely
confounders too
Ethical issues – the wider aspects
what information to give before seeking consent?deviation from normal clinical practice?
what full burden will be imposed on participants?
what risks will participants/others be exposed to?
what benefit might participants or others receive?
how might society/future patients benefit in time?
might publication reveal patients’ identities?
Exactly what are you planning to do?
PICO
P - who are the patients or what’s the problem?
I - what is the intervention or exposure?C – what is the comparison group?
O - what is the outcome or endpoint?
More on PICO
Patientsdisease or condition
stage, severity
demographic characteristics (age, gender, etc.)
Intervention
type of intervention or exposure
dose, duration, timing, route, etc.
Comparison
risk or treatment
placebo or other active treatment
Outcome
frequency, risk, benefit, harm
dichotomous or continuous
type: mortality, morbidity, quality of life, etc.
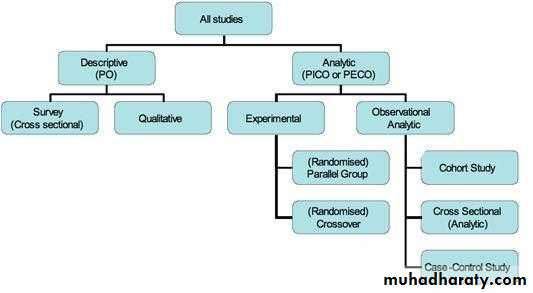
Study designs
Population (P) Outcomes (O)
Interventions (I) or Exposures (E)Centre for Evidence Based Medicine, Oxford, UK www.cebm.net
Are you going to observe or experiment?
observational – cross sectional, case series, case-control studies,cohort studies
identify participants
observe and record characteristics
look for associations
experimental – before and after studies, comparative trials
(controlled or head to head), randomised trials (ditto)identify participants
place in common context
intervene
observe/evaluate effects of intervention
Pros and cons of the RCT
An experimental comparison study where participants are allocated to
treatment/intervention or control/placebo groups using a random
mechanism. Best for studying the effect of an intervention.
Advantages:
unbiased distribution of confoundersblinding more likely
randomisation facilitates statistical analysis
Disadvantages:
expensive: time and money
volunteer bias
ethically problematic at times
Pros and cons of crossover trial
A controlled trial where each participant has both therapiese.g is randomised to treatment A first then starts treatment B.
Advantages:
all participants serve as own controls and error variance is reduced, thus reducing sample size neededall participants receive treatment (at least some of the time)
statistical tests assuming randomisation can be used
blinding can be maintained
Disadvantages:
all participants receive placebo or alternative treatment at some point
washout period lengthy or unknown
cannot be used for treatments with permanent effects
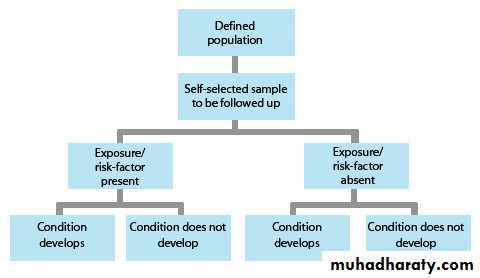
Pros and cons of cohort study
Data obtained from groups who have already been exposed, or not
exposed, to the factor of interest. No allocation of exposure is made by the
researcher. Best for studying effects of risk factors on an outcome.
Advantages:
ethically safe
participants can be matched
can establish timing and directionality of events
eligibility criteria and outcome assessments can be standardised
Disadvantages:
controls may be difficult to identify
exposure may be linked to a hidden confounder
blinding is difficult
for rare disease, large sample sizes or long follow-up necessary
Cohort study
Chronic kidney disease and riskof major cardiovascular disease
and non-vascular mortality:
Prospective population based
cohort study.
Di Angelantonio E, et al.
BMJ 341:doi:10.1136/bmj.c4986
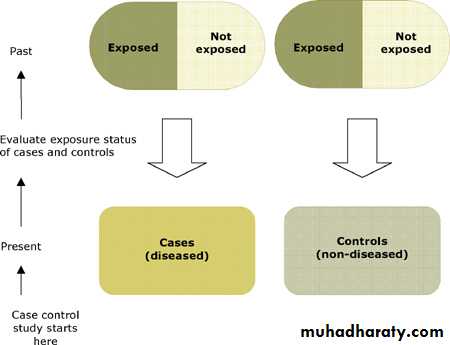
Pros and cons of case-control study
Patients with a certain outcome or disease and an appropriate group ofcontrols, without the outcome or disease, are selected (usually with some
matching) then information is obtained on whether the subjects have been
exposed to the factor under investigation.
Advantages:
quick and cheap as fewer people needed than cross-sectional studies
only feasible method for very rare disorders or those with long lag between exposure and outcome
Disadvantages:
reliance on recall or records to determine exposure status
confounders
selection of control groups is difficult
potential bias: recall, selection
Case-control study
Effectiveness of rotavirus
vaccination against childhood
diarrhoea in El Salvador:
case-control study.
de Palma O et al.
BMJ 340:doi:10.1136/bmj.c2825Pros and cons of cross sectional study
Examines the relationship between 1) diseases/other healthrelated characteristics and 2) other variables of interest as they exist in a
defined population at one time. Exposure and outcomes both measured at
the same time. Quantifies prevalence, risk, or diagnostic test accuracy
Advantages:
cheap and simple
ethically safe
Disadvantages:
establishes association at most, not causality
recall bias, social desirability bias
researcher’s (Neyman) bias
group sizes may be unequal
confounders may be unequally distributed
Cross sectional study
Sociodemographic patterning of
non-communicable disease riskfactors in rural India: a cross
sectional study.
Kinra S et al.
BMJ 341:doi:10.1136/bmj.c4974Reporting statements
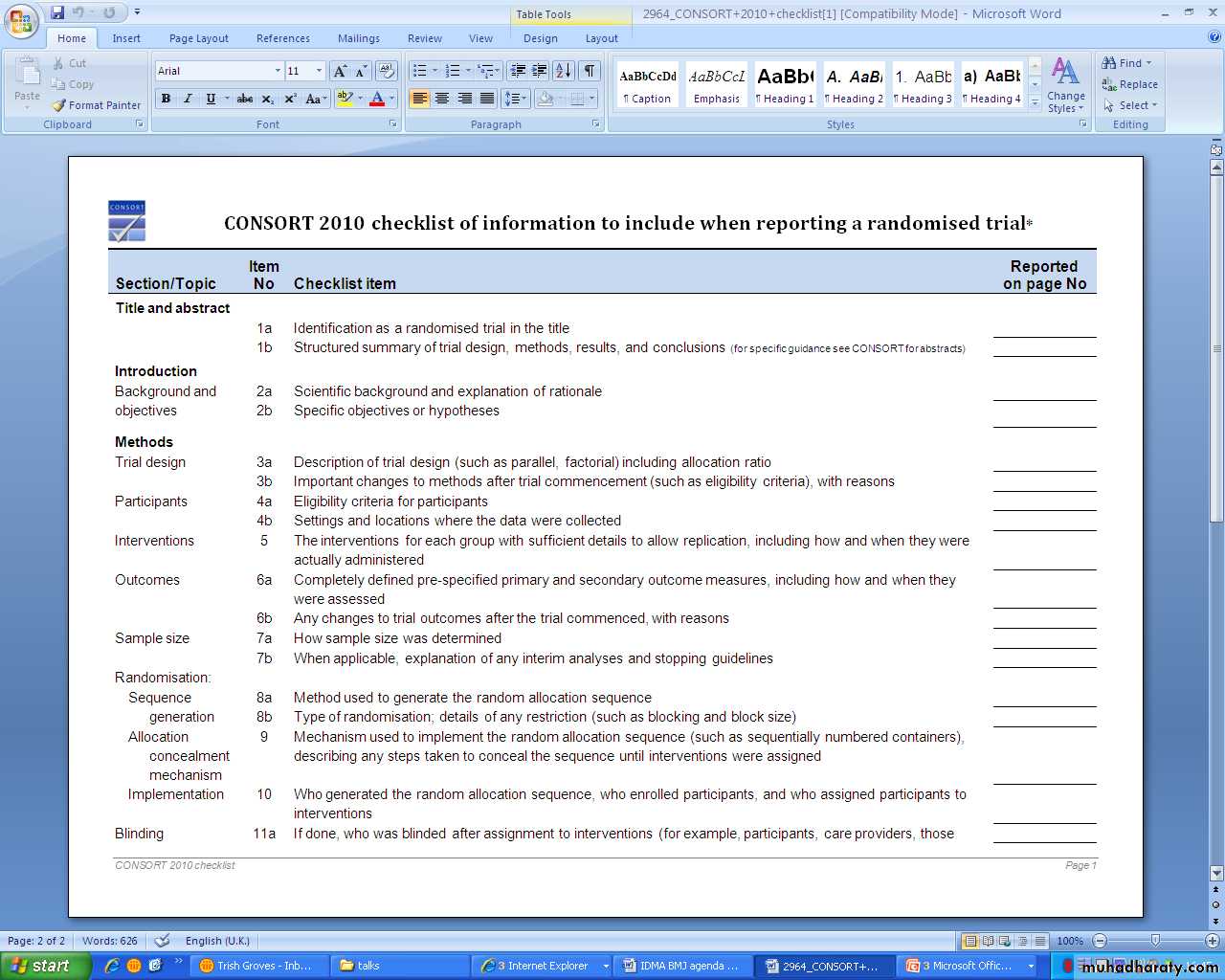
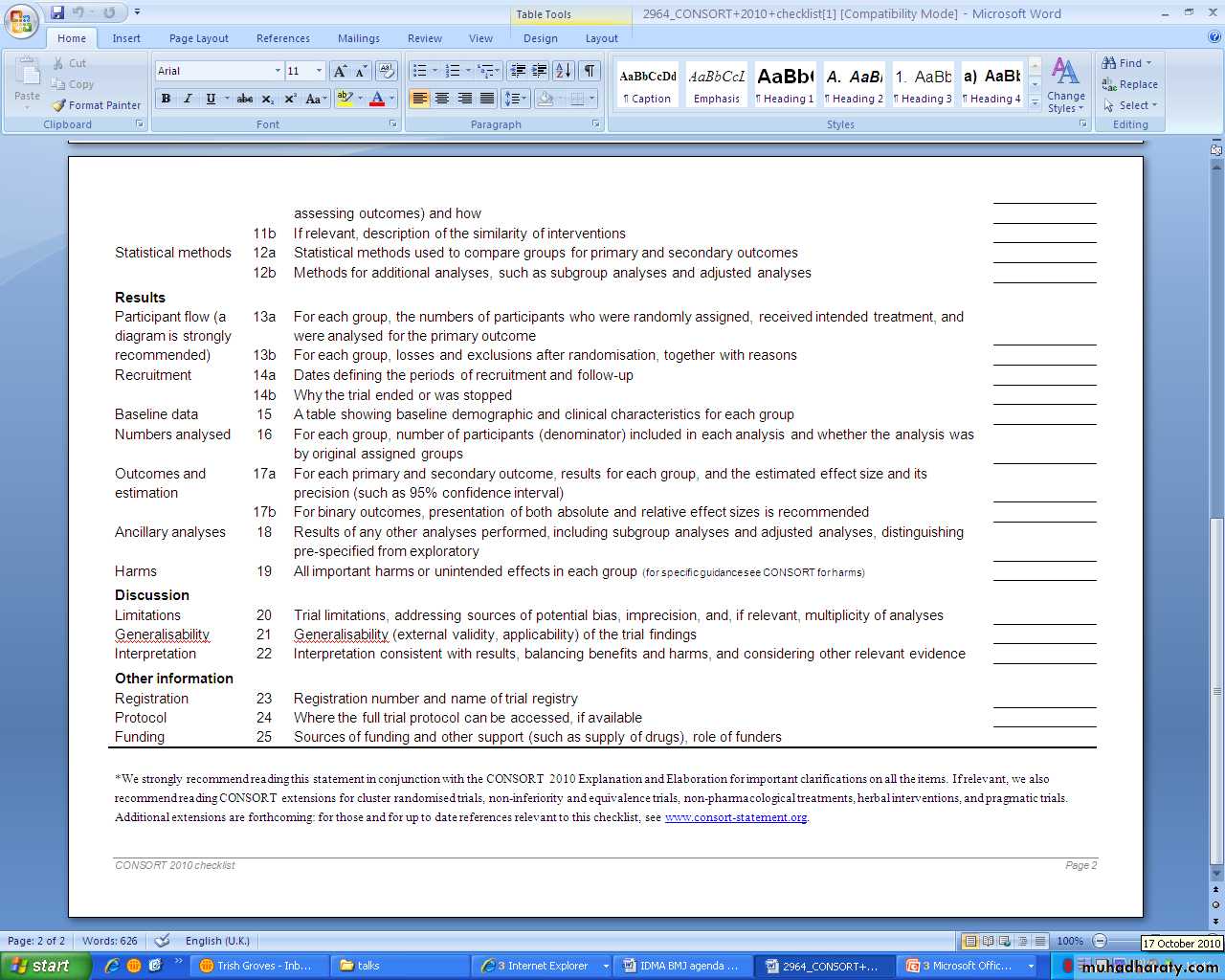
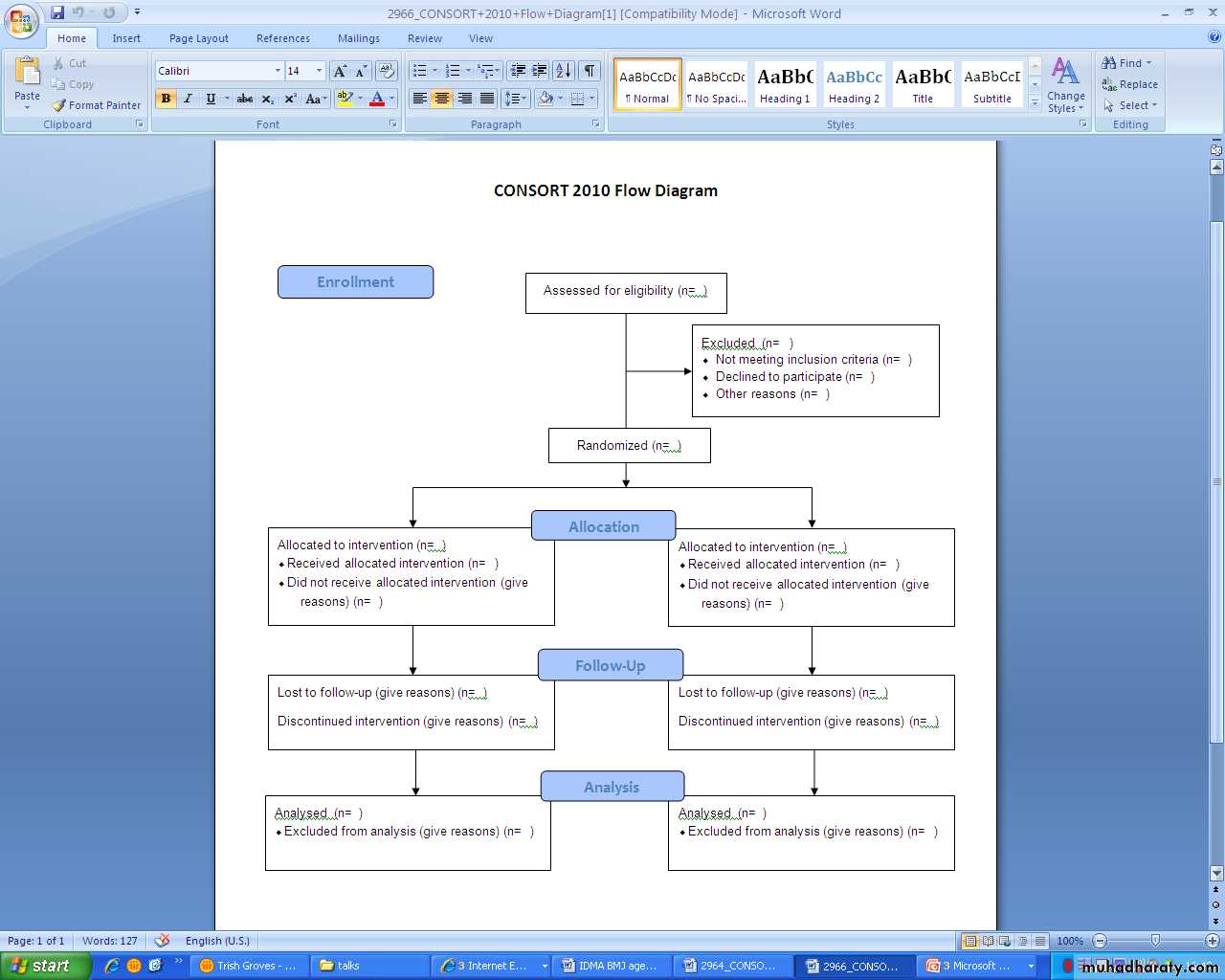
CONSORT for randomised controlled trialsSTARD for diagnostic accuracy studies
STROBE for observational studies
PRISMA for systematic reviews of trials
MOOSE for meta-analyses of observational studies
EQUATOR network
equator-network.org/resource-centre/library-of-health-research reporting/CONSORT 2010CONsolidated Standards of Reporting Trials