
Chapter 5
Third Week of
Development:
Trilaminar Germ Disc
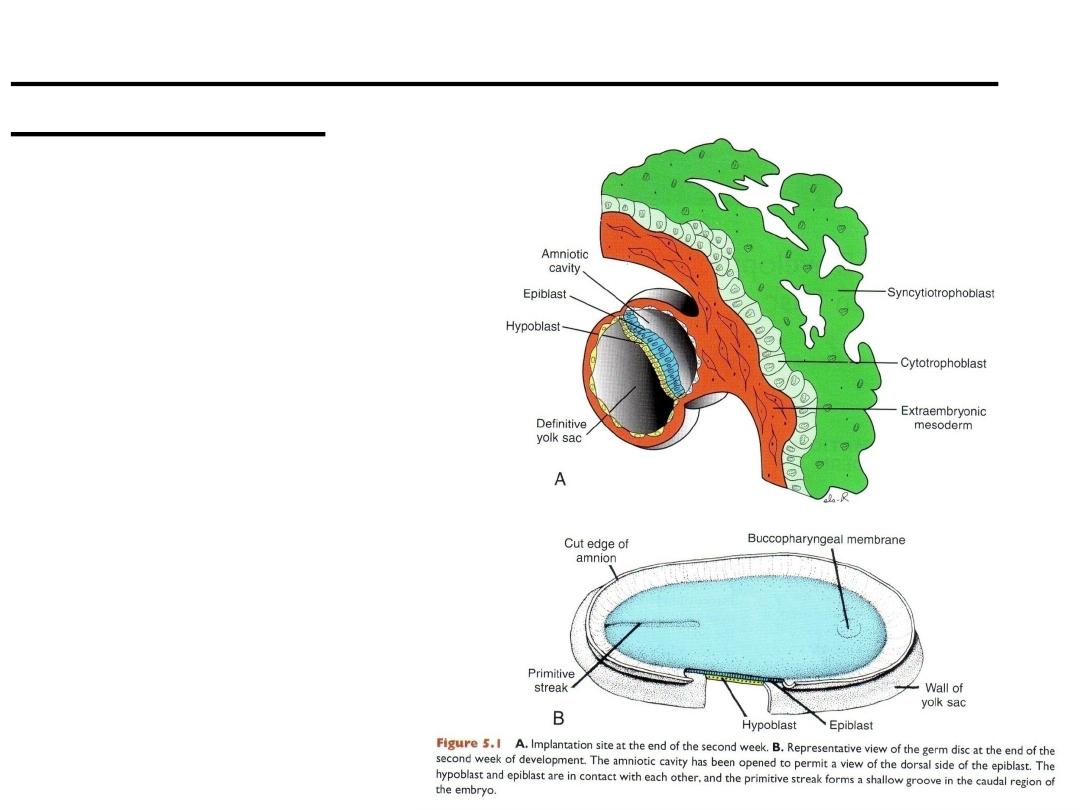
A. Implantation site at the end
of the second week.
B. Representative view of the
germ disc at the end of the
second week of development.
The amniotic cavity has been
opened to permit a view of the
dorsal side of the epiblast. The
hypoblast and epiblast are in
contact with each other, and
the primitive streak forms a
shallow groove in the caudal
region of the embryo.
Gastrulation (Formation Of Embryonic Mesoderm
And Endoderm)

Gastrulation
• Gastrulation: Third week of gestation
– Process that establishes all three germ layers (ectoderm, mesoderm, and
endoderm).
– Begins with formation of the primitive streak on the surface of the epiblast.
• The primitive node surrounds the small primitive pit.
• Invagination: cells of the epiblast migrate toward the primitive streak and slip
beneath it.
• Controlled by fibroblast growth factor 8 (FGF8),
• Once the cells have invaginated, some displace the hypoblast, creating the
embryonic endoderm, and others come to lie between the epiblast and newly
created endoderm to form mesoderm.
• Cells remaining in the epiblast then form ectoderm.
• Thus, the epiblast, through the process of gastrulation, is the source of all of the
germ layers.
• cells in these layers will give rise to all of the tissues and organs in the embryo.
• They contact with the extra embryonic mesoderm covering the yolk sac and
amnion.
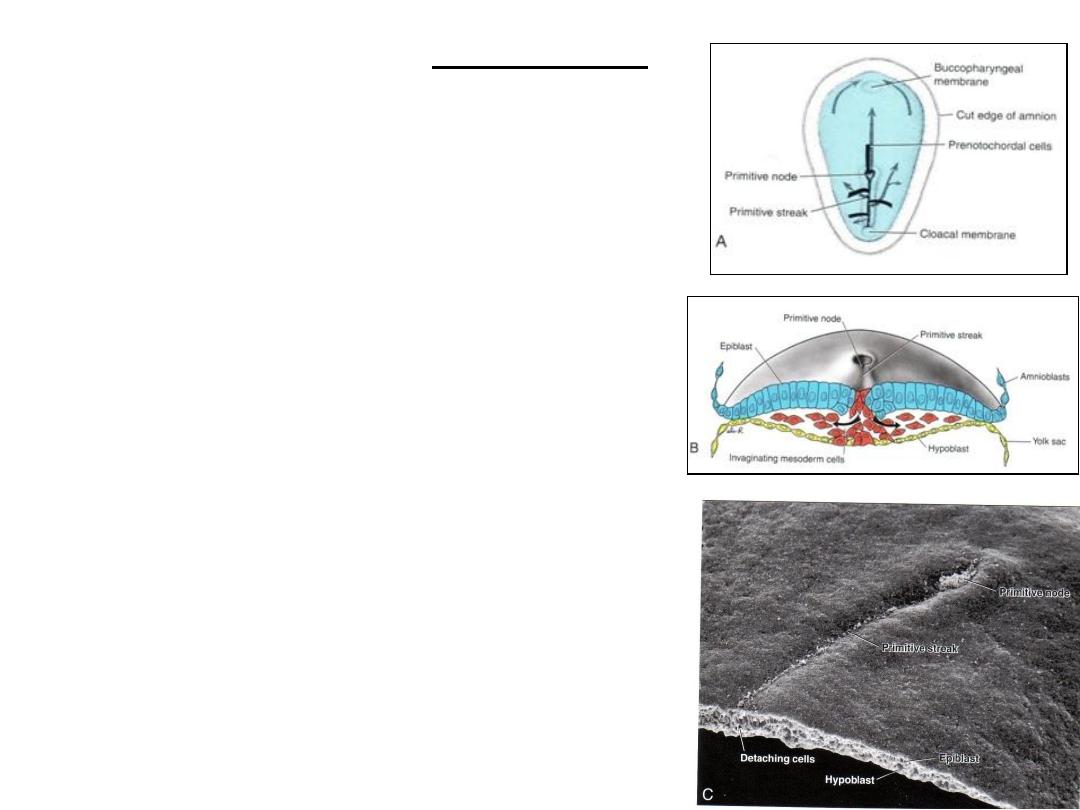
Invagination
A. Dorsal side of the germ disc from a 16-day embryo
indicating the movement of surface epiblast cells (solid
black lines) through the primitive streak and node and
the subsequent migration of cells between the
hypoblast and epiblast (broken lines).
B. Cross section through the cranial region of the
streak at 15 days showing invagination of epiblast
cells. The first cells to move inward displace the
hypoblast to create the definitive endoderm.
Once definitive endoderm is established, inwardly
moving epiblast forms mesoderm.
C. Dorsal view of an embryo showing the primitive
node and streak and a cross section through the
streak. Arrow, detaching epiblast cells in the
primitive streak.
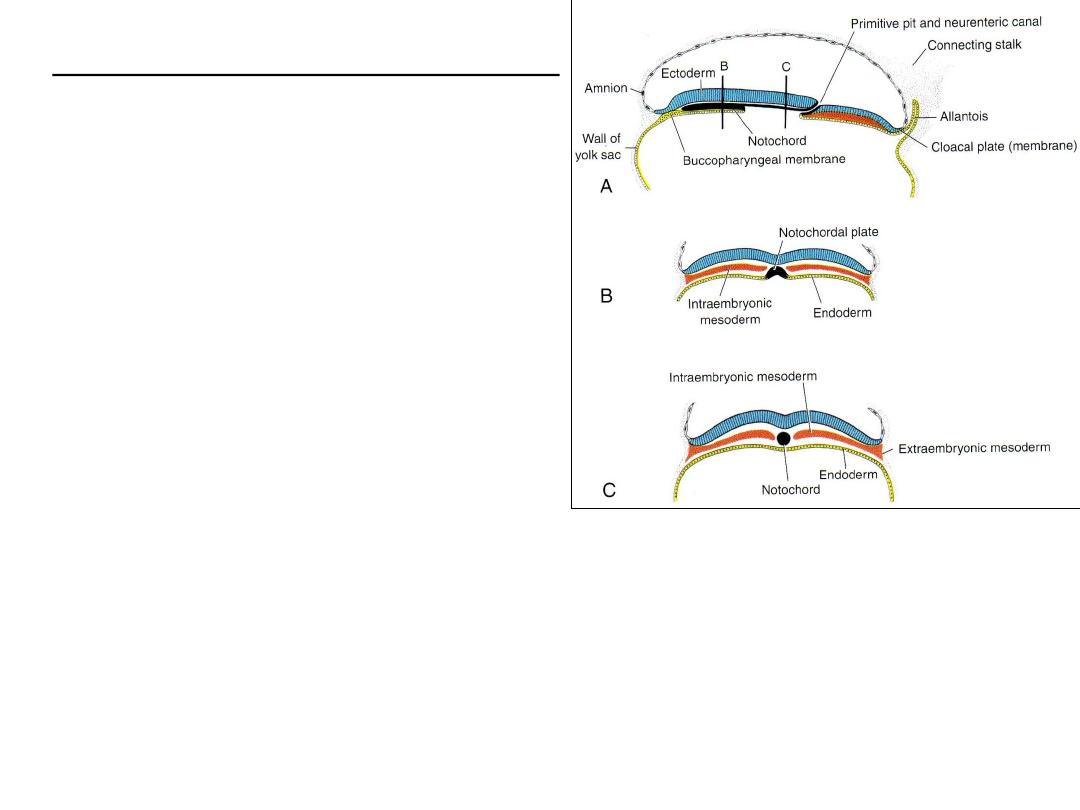
FORMATION OF THE NOTOCHORD
•
Prenotochordal cells invaginate in primitive
node, reach prechordal plate
•
Prenotochordal cells + hypoblast =
notochordal plate.
•
Notochordal plate then forms a solid cord of
cells, the definitive notochord
•
The notochord underlies the neural tube and
serves as the basis for the axial skeleton.
•
The notochord and prenotochordal cells
extend cranially to the prechordal plate (an
area just caudal to the oropharyngeal
membrane) and caudally to the primitive pit.
•
The oropharyngeal membrane at the cranial end of the disc consists of a small region of
tightly adherent ectoderm and endoderm cells that represents the future opening of
the oral cavity
•
The cloacal membrane is formed at the caudal end of the embryonic disc consists of
tightly adherent ectoderm and endoderm cells with no intervening mesoderm.
•
The allantoenteric diverticulum, or allantois: from the posterior wall of the yolk sac,
extends into the connecting stalk.
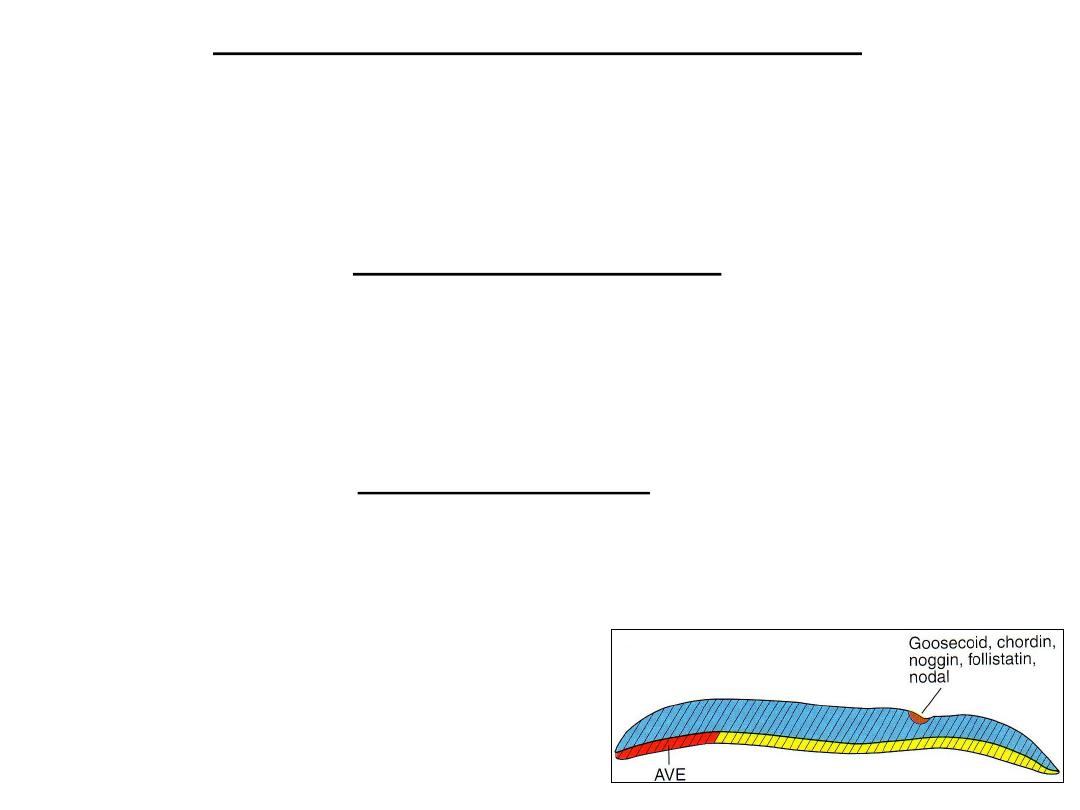
ESTABLISHMENT OF THE BODY AXESE
•
Body axes, antero-posterior, dorso-ventral, and left–right, takes place before and during the
period of gastrulation.
•
Sagittal section through the node and primitive streak showing the expression pattern of
genes regulating the cranio- caudal and dorso- ventral axes.
Antero-posterior axis
•
Cells at the prospective cranial end of the embryo in the anterior visceral endoderm
(AVE) express the transcription factors OTX2, LIM1,and HESX1and the secreted factor
cerberus and lefty which inhibit nodal activity in the cranial end of the embryo. These
genes establish the cranial end of the embryo before gastrulation.
Dorso-ventral axis
• The node is the ORGANIZER (nodal).
• Nodal is involved in initiating and maintaining
the primitive streak.
• Once the streak is formed and gastrulation is
progressing, bone morphogenetic protein
(BMP4; hatched areas), secreted throughout
the bilaminar disc, acts with FGF to ventralize
mesoderm into kidneys (intermediate
mesoderm) and blood and body wall
mesoderm (lateral plate mesoderm).

The Node Is The Organizer.
• All mesoderm would be ventralized if the activity of BMP4 were not
blocked by other genes expressed in the node. For this reason the node
is the organizer.
• These genes are: Chordin (activated by the transcription factor
Goosecoid), noggin and follistatin antagonizes the activity of BMP4.
• As a result, cranial mesoderm is dorsalized into notochord , somites and
somitomeres in the head region.
Goosecoid gene
• Because Goosecoid activates inhibitors of BMP4 and regulates head
development, over or under expression of this gene results in severe
malformation in the head region: as duplications of head as in some types
of conjoined twins.

Conjoined twins
• If the gene Goosecoid is overexpressed in frog
embryos, the result is a two-headed tadpole.
Perhaps, overexpression of this gene explains the
origin of this type of conjoined twins
Brachyury (T) gene
• Regulation of dorsal mesoderm formation in middle and caudal regions is
controlled by the Brachyury (T) gene expressed in the node.
• Absence of Brachyury (T) gene results in shortening of the embryonic axis
(caudal dysgenesis).
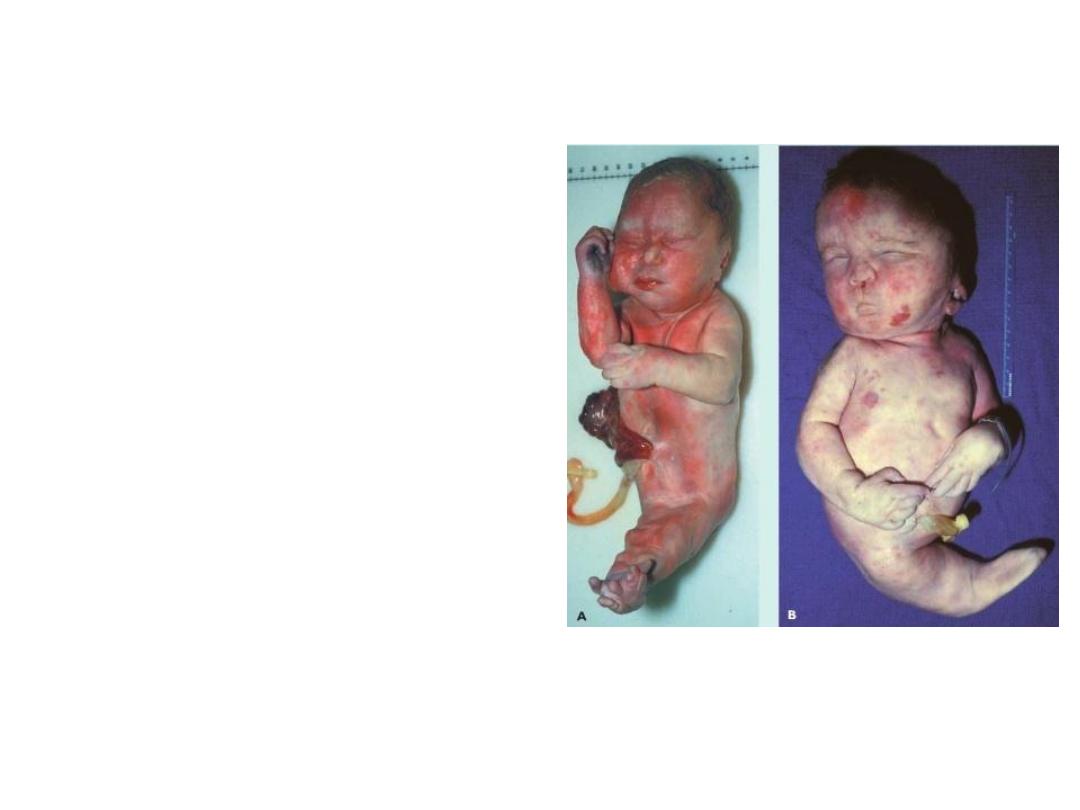
Caudal dysgenesis (sirenomelia)
•Insufficient mesoderm is formed in the
caudal most region of the embryo.
Because this mesoderm contributes to
formation of the lower limbs, urogenital
system (intermediate mesoderm), and
lumbosacral vertebrae, abnormalities in
these structures ensue. Affected
individuals exhibit a variable range of
defects, including hypoplasia and fusion
of the lower limbs, vertebral
abnormalities, renal agenesis,
imperforate anus, and anomalies of the
genital organs.
•In humans, the condition is associated
with maternal diabetes and other
causes.
•In mice, abnormalities of Brachyury(T)
produce a similar phenotype.
A, B. Two examples of sirenomelia (caudal
dysgenesis). Loss of mesoderm in the
lumbosacral region has resulted in fusion
of the limb buds and other defects.
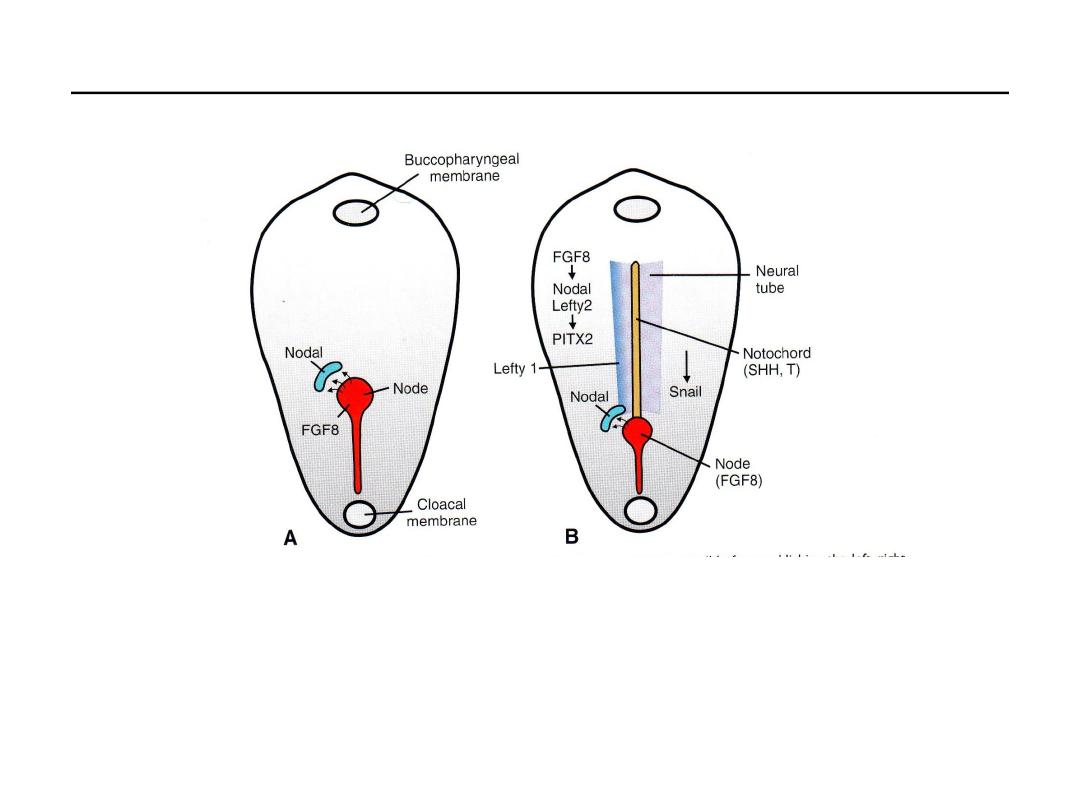
Gene expression patterns responsible for left-right body axis
• Left–right asymmetry is regulated by a cascade of genes; first, FGF8,
secreted by cells in the node and streak, induces Nodal and LEFTY-2
expression on the left side.
• These genes up-regulate PITX2, a transcription factor responsible for left-
sidedness.
• Heart, stomach and other gut primordia.
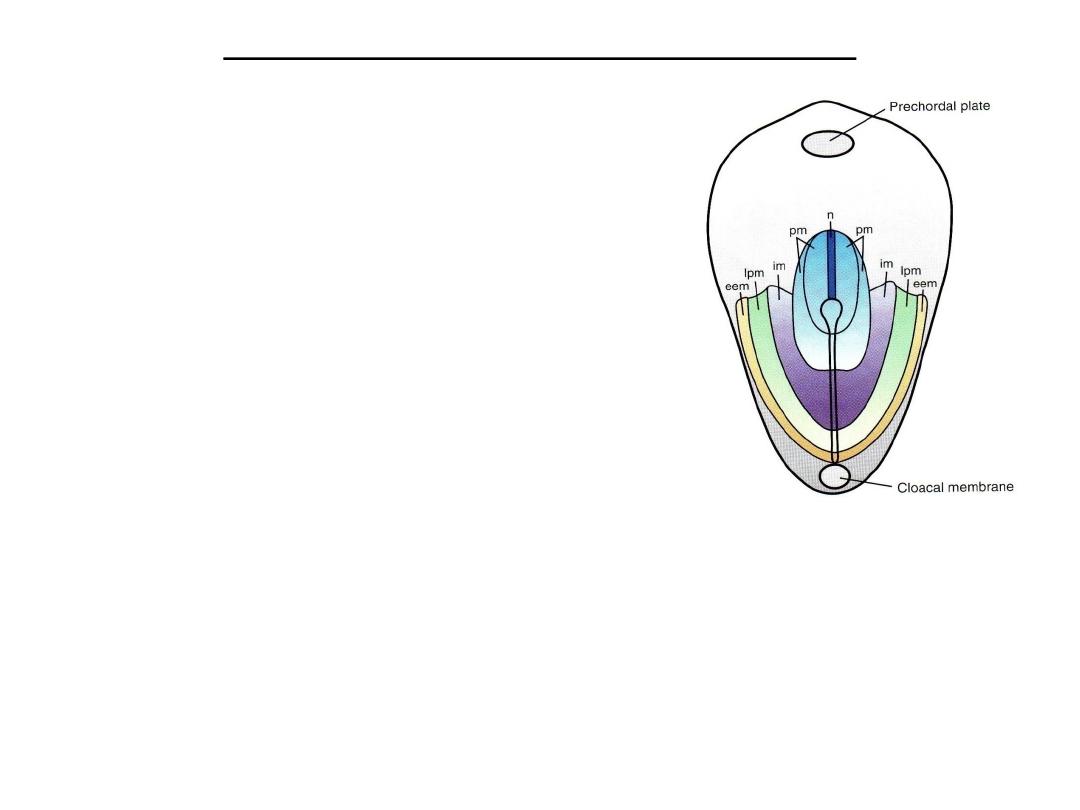
Fate map established during gastrulation
• Epiblast cells moving through the node and
streak are predetermined by their position
to be-come specific types of mesoderm and
endoderm. Thus, it is possible to construct a
fate map of the epiblast showing this pattern
.
Dorsal view of the germ disc showing the primitive streak and a fate map for epiblast cells.
Specific regionsof the epiblast migrate through different parts of the node and streak to form
mesoderm. Thus, cells migrating at the cranial most part of the node will form the notochord
(n); those migrating more posteriorly through the node and cranial most aspect of the streak
will form paraxial mesoderm (pm;somitomeres and somites); those migrating through the
next por-tion of the streak will form intermediate mesoderm (im;urogenital system); those
migrating through the more caudal partof the streak will form lateral plate mesoderm
(lpm;body wall); and those migrating through the most caudal part will contribute to
extraembryonic mesoderm (eem;chorion)

Teratogenesis associated with gastrulation
• Beginning of 3
rd
wk:
• Gastrulation
• Sensitive to teratogenes: fate map to organ systems: eyes & brain
• Alcohol: HOLOPROSENCEPHALY
• Genetic abnomalities:
– caudal dysgenesis (sirenomelia):
• maternal diabetes
• abnormalities of Brachyury (T)
Situs inversus
(complete)
• A condition in which transposition of the viscera in the thorax and
abdomen occurs.
• With few other structural abnormalities
• With bronchiectasis and chronic sinusitis
– because of abnormal cilia (Kartagener syndrome).
Laterality sequences
• do not have complete situs inversus
• predominantly bilaterally left-sided or right-sided
• The spleen reflects the differences
• left-sided bilaterality have polysplenia
• right-sided bilaterality have asplenia or hypoplastic spleen.
• Patients with laterality sequences are also likely to have other
malformations, especially heart defects.
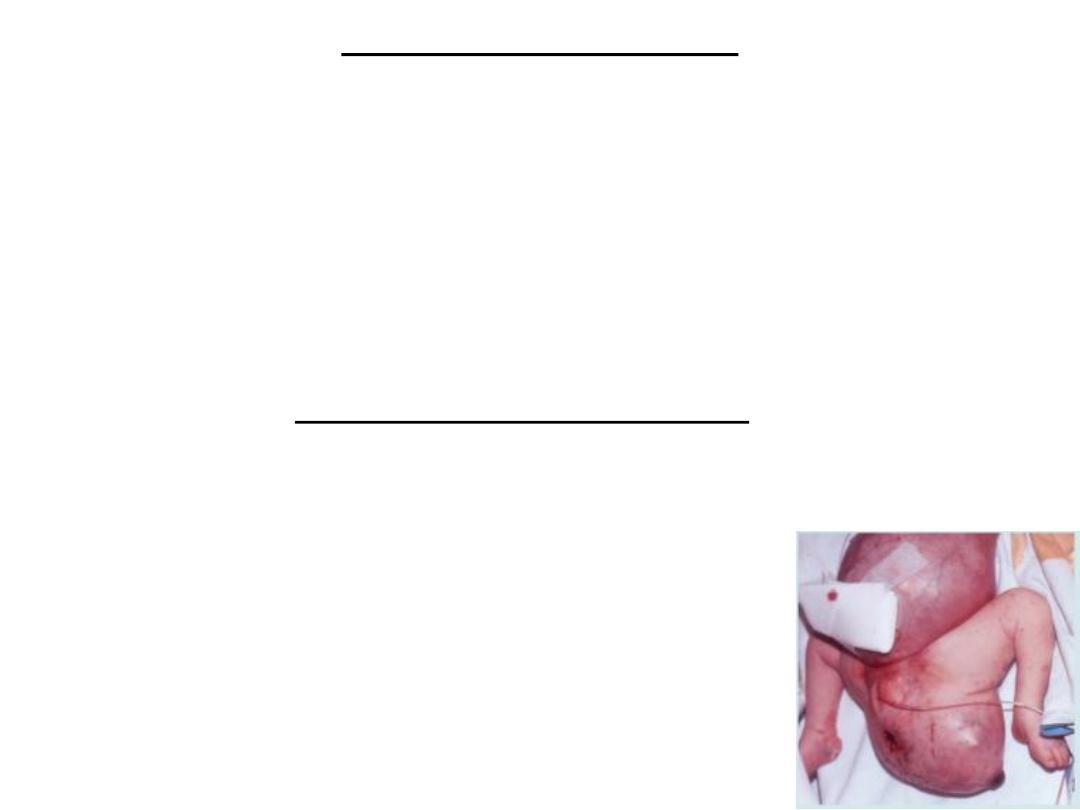
Sacrococcygeal teratoma
• Tumors Associated With Gastrulation
• remnants of the primitive streak persist in the
sacrococcygeal region. These clusters of pluripotent cells
proliferate and form tumors, known as sacrococcygeal
teratomas, that commonly contain tissues derived from all
three germ layers.
• This is the most common tumor in newborns, occurring
with a frequency of one in 37,000. These tumors may also
arise from primordial germ cells that fail to migrate to the
gonadal ridge.
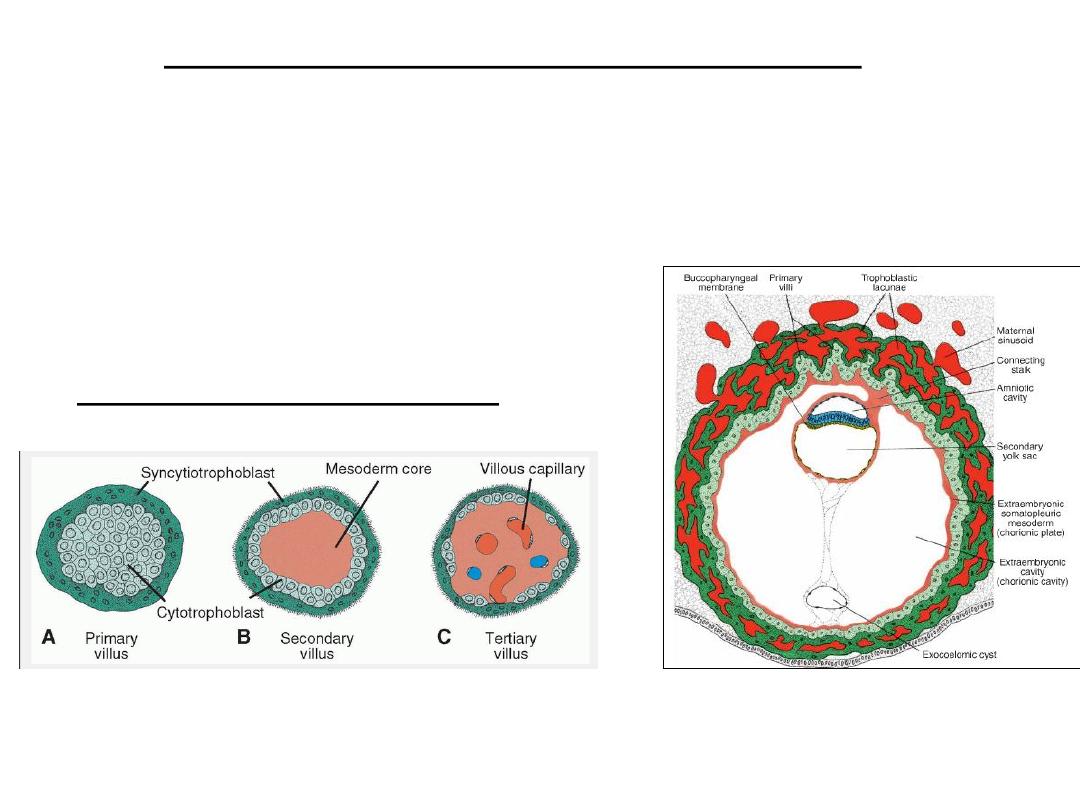
Further development of the trophoblast
• The trophoblast progresses rapidly.
• Primary villi obtain a mesenchymal core in which small capillaries arise.
When these villous capillaries make contact with capillaries in the
chorionic plate and connecting stalk, the villous system is ready to supply
the embryo with its nutrients and oxygen
A 13-day-old implantation site showing
primary villi of the trophoblastic shell
just beginning to be invaded by
mesoderm from the chorionic plate.
Development of a villus.
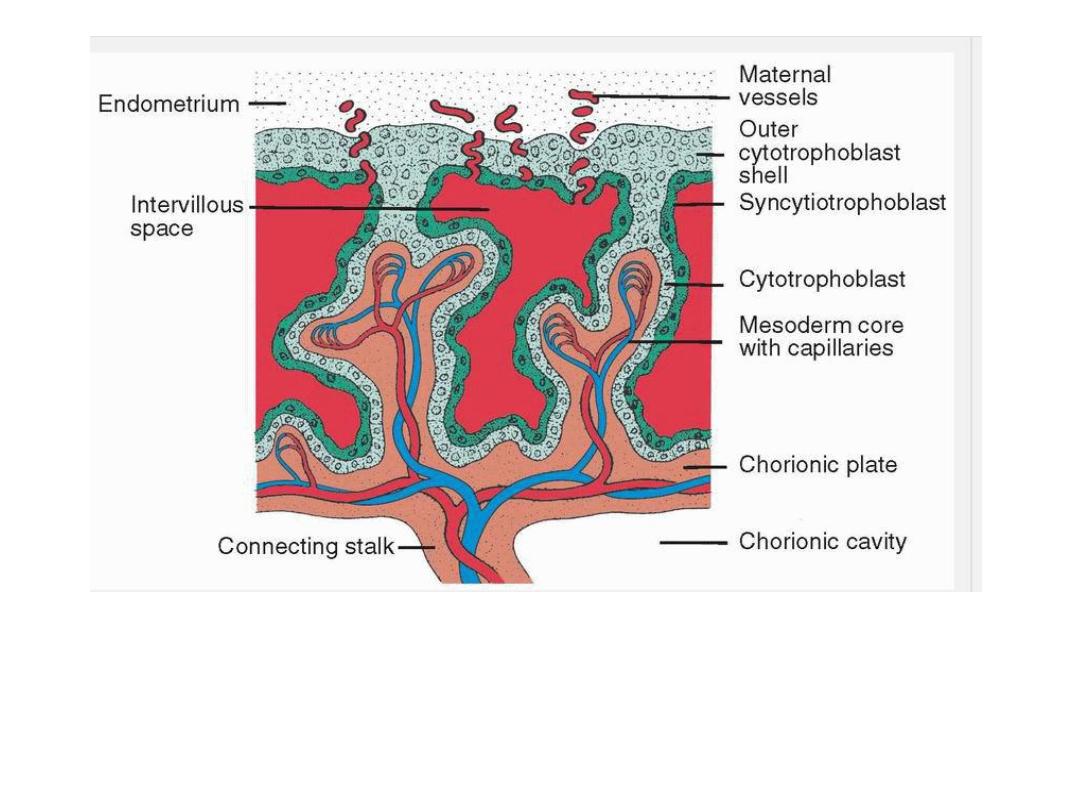
Longitudinal section through a villus at the end of the third week of development.
Maternal vessels penetrate the cytotrophoblastic shell to enter intervillous spaces,
which surround the villi. Capillaries in the villi are in contact with vessels in the
chorionic plate and in the connecting stalk, which in turn are connected to intra
embryonic vessels
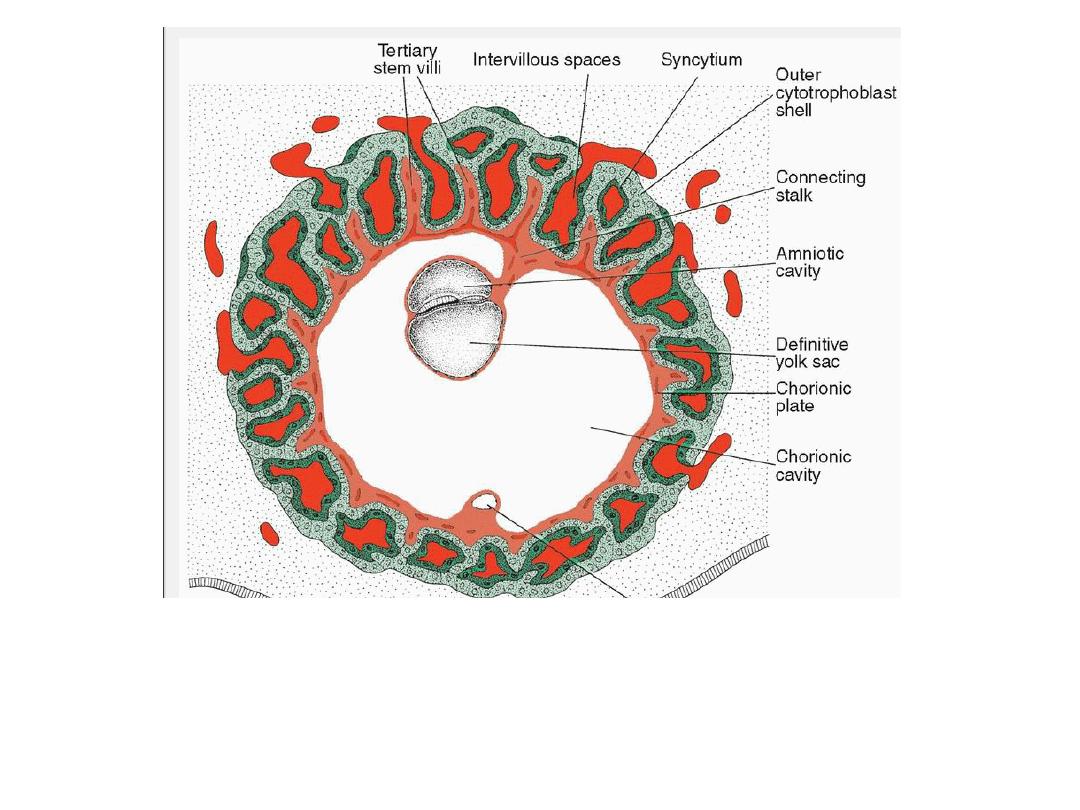
Presomite embryo and the trophoblast at the end of the third week. Tertiary and
secondary stem villi give t he trophoblast a characteristic radial appearance.
Intervillous spaces, which are found throughout the trophoblast, are lined with
syncytium. Cytotrophoblastic cells surround the trophoblast entirely and are in direct
contact with the endometrium. The embryo is suspended in the chorionic cavity by
means of the connecting stalk
