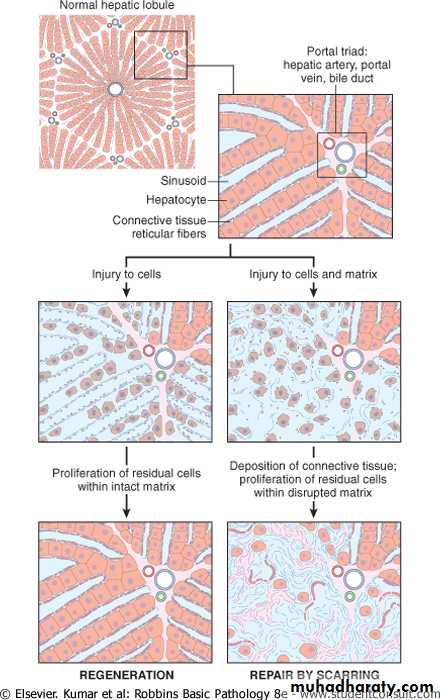
Regeneration:- complete restitution of the damaged components of the affected tissue i.e. the tissue essentially return to normal state.
Healing:- is a reparative process characterized by laying down of connective (fibrous) tissue that result in scar formation, this mode occurs when
The injured tissues are incapable of complete regeneration or
The supporting structures of the tissue are severely damaged
Although the resulting fibrous scar is not normal, it provides enough structural stability that allows the injured tissue to function.
Therefore an understanding of the process of repair requires some knowledge of:-
• the control of cell proliferation• the activity of growth factor
• the functions of the ECM.
• and the interactions between cells and ECM components.
The control of cell proliferation
Several cell types proliferate during tissue repair, these include
• The remnants of the injured tissue (which attempt to restore normal structure)
• Vascular endothelial cells (to create new vessels that provide the nutrients for the repair process)
• Fibroblasts (the source of the fibrous tissue that fills defects)
The proliferation of the above cell types is driven by growth factors. The production of polypeptide growth factors, responses of cells to these factors, and the ability of these cells to divide and expand in numbers are all important determinant of the adequacy of the repair process
The cell cycle
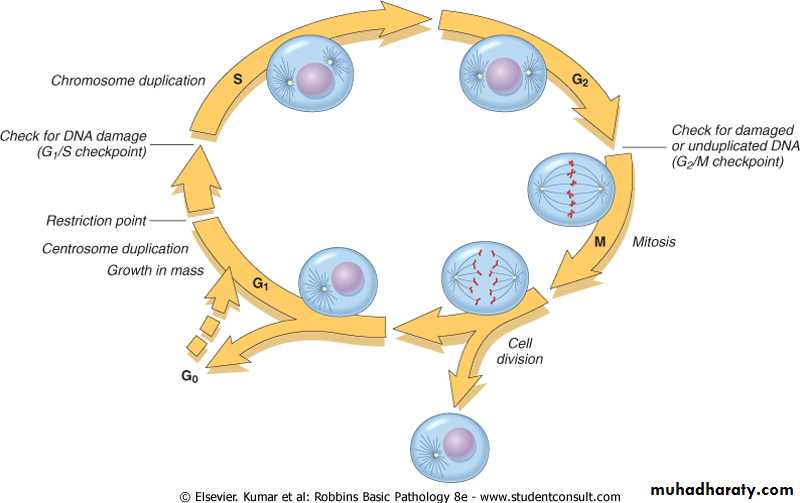
The cell cycle represents the sequence of events that control the DNA replication & mitosis in the proliferation of cells. It consist of a series of steps at which the cell checks for the accuracy of the process and instructs itself to proceed to the next step.The cycle consist of the presynthetic growth phase 1 (G1), the DNA synthesis phase (s), the premitotic growth phase 2 (G2), and the mitotic phase (M).
non dividing cells are either in cell cycle arrest in G1 or they exit the cycle to enter a phase called G0.
Any stimulus that initiate the cell proliferation such as exposure to growth factors, need to promote the G0/G1 transition and the entry of cells into the G1.
Further progression is determined by the ability of the cell to pass through the intrinsic quality control mechanism for cell integrity known as checkpoint control.
Checkpoint controls prevent DNA replication or mitosis of damaged cells and either transiently stop the cell cycle to allow for DNA repair or eliminate irreversibly damaged cell by apoptosis.
Progression through the cell cycle from G1 is regulated by proteins called cyclins, which form complexes with enzymes called cyclin- dependent kinases (CDKs). These complexes regulate the phosphorylation of proteins involved in cell cycle progression leading to DNA replication and mitosis and thus are required for cell cycle progression.
Stages of cell cycle
Presynthetic (G1) and synthetic (S) stages generally constitute the majority of the time of the cell cycle; the mitotic (M) phase is typically briefProliferative capacities of tissuesTissue repair is critically influenced by the intrinsic proliferative capacity of the constituent cells. Based on this criterion, this tissue of the body are divided into three groups:-
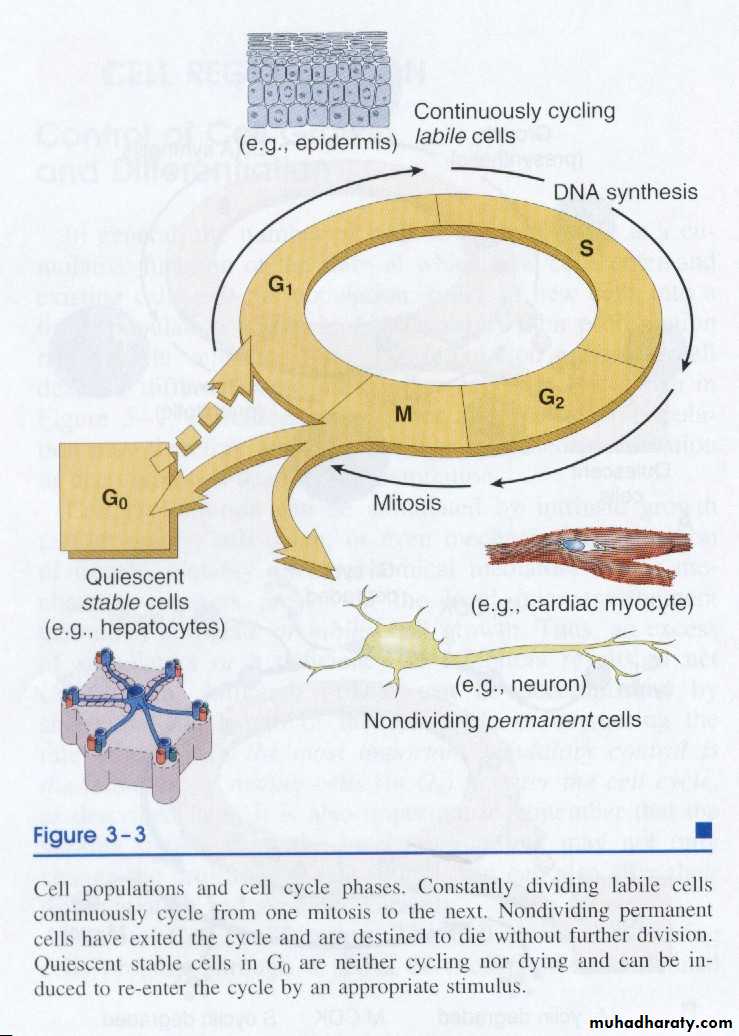
Continuously dividing tissues (labile tissues):- cells of these tissues are continuously being lost and replaced by maturation from stem cells and by proliferation of mature cells. Labile cells include hematopoietic cells in the bone marrow and the majority of surface epithelia.
Stable tissues:- cells of these tissues are quiescent (in the G0 of the cell cycle) and have only minimal replicative activity in their normal state. However these cells are capable of proliferating in response to injury or loss of tissue mass. Stable cells constitutes the parenchyma of most solid tissues, such as liver & kidney. They also include the endothelial cells, fibroblast and smooth muscle cells; the proliferation of these cells is particularly important in wound healing, with the exception of liver stable tissue have a limited capacity to regenerate after injury.
Cell types and cell cycle phases
Constantly dividing labile cells continuously cycling from one mitosis to the next. Nondividing permanent cells have exited the cycle and are distended to die without further division. Quiescent stable cells in G0 are neither cycling nor dying and can be induced to re-enter the cycle by an appropriative stimulus.
Permanent tissue:- cells of these tissues are terminally differentiated and non proliferative in postnatal life. The majority of the neurons and cardiac muscle cells belong to this category. Accordingly injury to the brain or heart is irreversible and result in scar. Skeletal muscle is usually classified as a permanent tissue, but satellite cells attached to the endomysial sheath provide some regenerative capacity for this tissue.
Growth factors
Cell proliferation can be triggered byGrowth factors
HormonesCytokines
Signals from the ECM
The polypeptide growth factors have a major role in cell survival
through protection from apoptotic death &stimulating cell division & proliferation.
also stimulate migration, differentiation and the synthesis of specialized proteins such as collagen in fibroblasts.
A major activity of growth factors is to stimulate the function of the growth control genes, many of which are proto-oncogenes (so named because mutations in them lead to unrestrained cell proliferation characteristic of neoplasia (oncogenes).
Many of the growth factors that are involved in repair are produced by leukocytes that are recruited & activated at the site of the injury, as part of the inflammatory process.
Other growth factors are produced by the specialized tissue (paranchymal) cells or the stromal (connective tissue) cells in response to cell injury or loss.
Signaling mechanism of growth factor receptors
the major intracellular signaling pathways induced by growth factor receptors that recognize extracellular ligands. The binding of a ligand to its receptor triggers a series of events by which extracellular signals are transduced into the cell leading to the stimulation or repression of gene expression.
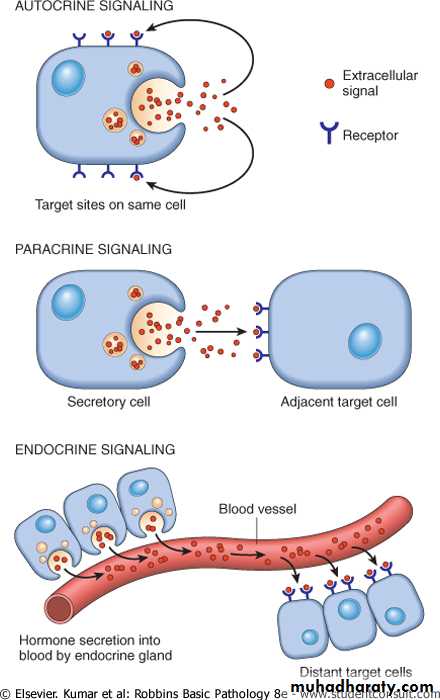
Signaling may occur directly in the same cell (autocrine signaling e.g. lymphocytes proliferation induced by cytokines in some immune responses),
between adjacent cells (paracrine signaling e.g. recruiting inflammatory cells to the site of the infection and in wound healing),
or overt great distances (endocrine signaling e.g. a hormone is released into the blood stream and act on the target cells at a distance).
Extracellular matrix and cell matrix interactions
Tissue repair depends not only on growth factor activity but also on interactions between cells and ECM components.The ECM is a dynamic constantly remodeling macromolecular complex synthesized locally,
which assembles into a network that surrounds cells.
It constitutes a significant proportion of any tissue.
By supplying a substrate for cell adhesion
and serving as a reservoir for growth factors.
ECM regulates the proliferation, movement and differentiation of the cells living within it.
Synthesis and degradation of ECM is associated with wound healing and chronic fibrotic processes.
Functions of the ECM:-
Scaffolding for tissue renewal: the maintenance of normal tissue structure requires a basement membrane or stromal scaffold. The integrity of the basement membrane or the stroma of the parenchymal cells is critical for the organized regeneration of tissues.It is particularly notable that although the labile and stable cells are capable for regeneration, injury to these tissues results in restitution of the normal structure only if the ECM is not damaged.
Disruption of these structures lead to collagen deposition and scar formation.
Establishment of tissue microenvironments: basement membrane act as a boundary between epithelium and underlying connective tissue and also form part of the filtration apparatus in the kidney.
ECM occur in two basic form:-
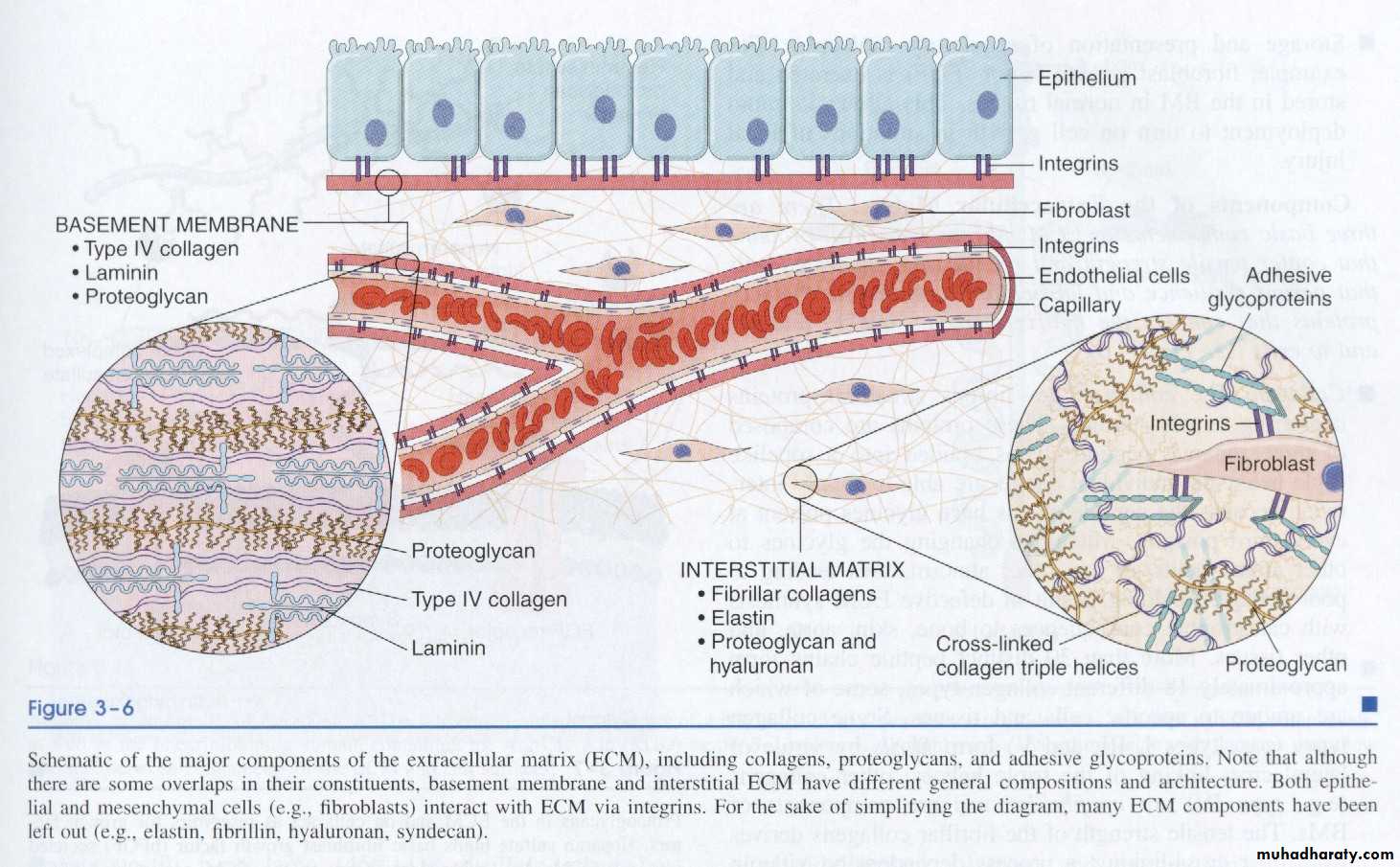
Interstitial matrix which is present in the spaces between mesenchymal (connective tissue) cells and between epithelium and supportive vascular and smooth muscle structures; it is synthesized by the mesenchymal cells (e.g. fibroblasts). Its major constituents are fibrilar and non fibrilar collagens as well as fibronectine, elastin, proteoglycans, hyaluronat and other elements.
Basement membrane, which lies beneath the epithelium and is synthesized by overlying epithelium and underlying mesenchymal cells; it tends to form a plate like mesh. Its major constituents are amorphous nonfibrilar type IV collagen and laminin.
Components of ECM
These include collagens, proteoglycans, and adhesive glycoproteins. Note that although there are some overlaps in their constituents, basement membrane and interstitial ECM have different general composition and architecture. Both epithelial and mesenchymal cells (e.g. fibroblasts) interact with ECM via integrins.
Component of the extracelluar matrix:-
There are three basic component of the ECM:Fibrous structural proteins (collagens and elastines) that confer tensile strength and recoil.
Water hydrate gels (proteoglycans and hyaluronan), which permit resilience and lubrication
Adhesive glycoproteins that connect the matrix element to one another and to cells.
Collagen:-
The collagen are fibrous structural proteins that give the tensile strength, without them human begins would be reduce to a clump of cells connected by neurons.Collagens are composed of three separated polypeptide chains braided into a ropelike triple helix. About 30 collagen types have been identified.
Some collagen types (e.g. type I,II, III,and V) form fibrils. The fibrillar collagen form a major proportion of the connective tissue in healing wound and particularly in scars.
The tensile strength of fibrillar collagens derive from their cross-linking. This process is dependent on vitamin C , therefore children with ascorbate deficiency have skeletal abnormalities, bleed easy because of weak vascular wall basement membrane and heal poorly.
Genetic defects in these collagenase causes diseases such as osteogenesis imperfecta and Ehler-Danols syndrome.
Other collagens are non fibrillar and may form basement membrane type IV or be a component of intervertebral discs (typeIX) or dermal-epidermal junction (type VII).
Elastin:-
The ability o the tissue to recoil and return to a baseline structure after physical stress is conferred by elastic tissue . this is especially important in the wall of the large vessels (which must accommodate recurrent pulsatile flow of the blood) as well as in the uterus, skin and ligaments.
Elastic fiber differed from collagen by having fewer cross –links. The fibrilin meshwork serve as a scaffold for the deposition of elastin and assembly of the elastic fibers ,
defect in the fibrilin synthesis lead to skeletal abnormalities and weakened aortic wall (Marfan syndrome)
Components of ECM
These include collagens, proteoglycans, and adhesive glycoproteins. Note that although there are some overlaps in their constituents, basement membrane and interstitial ECM have different general composition and architecture. Both epithelial and mesenchymal cells (e.g. fibroblasts) interact with ECM via integrins.
Proteoglycans and hyaluronan
Proteoglycans form highly hydrated compressible gels conferring resilience and lubrication ( such as in the cartilage in joints). They consist of long polysaccharides called glycosaminoglycans linked to a protein backbone.Hyaluronan, a huge molecule composed of many disaccharide repeats without a protein core , is also an important constituent of the ECM. Because its ability to bind to water, it form a viscous, gelatin like matrix. Besides providing compressibility to a tissue, proteoglycans can also be integral cell membrane protiens and have roles in cell proliferation, migration and adhesion.
Adhesive glycoproteins and adhesion receptors:
They are structurally diverse molecules involved in cell to cell adhesion, the linkage between the cells and the ECM, and binding between ECM components. The adhesive glycoproteins include fibronectin (major component of the interstitial ECM ) and laminin (major constituent of the basement membrane ).
The adhesion receptors, also known as cell adhesion molecules (CAMS) are grouped into four families:
Immunoglobulines
Cadherines
Selectins
Integrins
Cell and tissue regeneration:-
Cell renewal occurs continuously in labile tissues such as the bone marrow, gut epithelium and skin. Damage to epithelia or an increased loss of blood cells can be corrected by the proliferation and differentiation of stem cells and,
.
Tissue regeneration can occur in parenchymal organs with stable cell populations, but with exception of the liver this is usually a limited process.
The surgical removal of the kidney elicits in the contralateral kidney a compensatory response that consists of both hypertrophy and hyperplasia of the proximal duct cells .
the regenerative response of the liver that occurs after surgical removal of hepatic tissue is striking. Up to 60% of the liver may be removed in a procedure called living –donor transplantation, in which a portion of the liver is resected from a normal individual and is transplanted in recipient with end stage liver disease, or after partial hepatectomies performed for tumor removal . in such cases the tissue resection triggers proliferation of the remaining hepatocytes (normal quiescent ).
Expermintally, hepatocytes replication after partial hepatectomy is initiated by cytokines (e.g tumor necrosis factors TNF and interleukin 6 (IL 6) . EGF (epidermal growth factor receptors, or EGFR) with intirinsic tyrosine kinase activity is mitogenic for hepatocytes
It should be emphasized that extensive regeneration or compensatory hyperplasia can occur only if the residual tissue is structurally and functionally intact , as after partial surgical resection . by contrast, if the tissue is damaged by infection or inflammation regeneration is incomplete and is accompanied by scaring.
Repair by connective tissue
Healing or repair by connective tissue is encountered if• A severe or chronic persistent tissue injury that result in damage to parenchymal cells as well as the stromal framework
• Injuries affects nondividing cells
Under these conditions, repair occur by replacement of the non-regenerated cells with the connective tissue, or by a combination of regeneration of some cells and scar formation.
Repair begin within 24 h of injury by the emigration of the fibroblasts and the induction of fibroblast and endothelial cell proliferation.
By 3 to 5 days a specialized type of tissue that is characteristic of healing called granulation tissue is apparent . the term granulation tissue derive from the pink soft granular gross appearance, such as that seen beneath the scab of a skin wound . it is microscopic appearances is characterized by proliferation of fibroblast and new thin –walled , delicate capillaries (angiogenesis), in a loose ECM granulation tissue then progressively accumulated connective tissue matrix. Eventually resulting in the formation of a scar which may remodel over time.
At high magnification, granulation tissue has capillaries,
fibroblasts, and a variable amount of inflammatory cells(mostly mononuclear, but with the possibility of some PMN's
being present
macrophages
lymphocytes
Plasma cells
fibroblastsRepair by connective tissue deposition consist of four sequential processes:
• Formation of new blood vessels (angiogenesis)• Migration and proliferation of fibroblasts
• Deposition of ECM (scar formation)
• Maturation and reorganization of the fibrous tissue (remodeling)
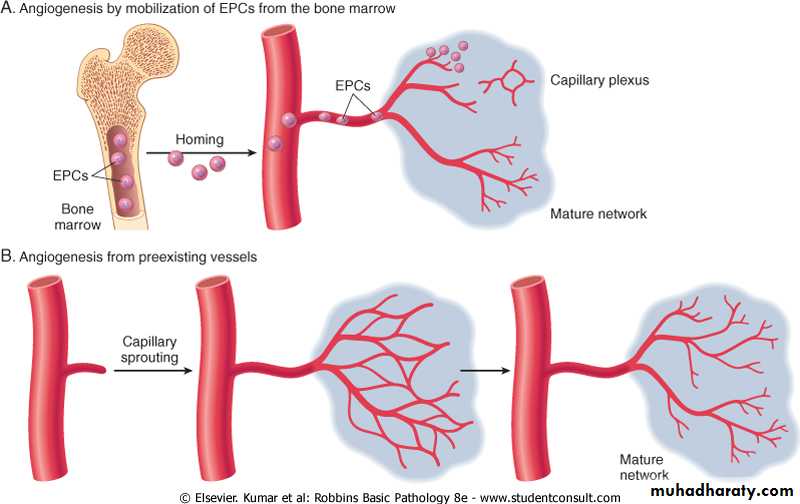
• Angiogenesis occur by two mechanism:-
The preexisting vessels send out capillary sprouts to produce new vessels. Angiogenesis is a critical process in healing at site of injury, in the development of the collateral circulation at site of ischemia and in allowing tumor to increase in size beyond the limits of their original blood supply.It has recently been found that endothelial precursor cells may migrate from the bone marrow to areas of injury and participate in angiogenesis at these sites .
New vessels formed during angiogenesis are leaky. This leakiness explains why granulation tissue is often edematous, and account in part for the edema that may persist in healing wounds long after the acute inflammatory response has resolved.
Several factor induce angiogenesis, but the most important are VEGF and basic fibroblast growth factor (FGF-2). VEGF stimulate both proliferation and motility of endothelial cells thus initiating the process of capillary sprouting . in angiogenesis involving endothelial cell precursors from the bone marrow and to induce proliferation and motility of these cells at the sites of angiogenesis.
Migration of fibroblasts and ECM Deposition (scar formation)
Scar formation builds on the granulation tissue framework. It occurs in tow steps:-Migration and proliferation of fibroblast into the site of the injury and
Deposition of ECM by these cells
The recruitment and stimulation of fibroblasts is driven by many growth factors including PDGF (platelet derived growth factor). One source of these factor is the activated endothelium but more importantly growth factors are also elaborated by inflammatory cells, macrophages in particular are important cellular constituents of granulation tissue, and beside clearing extracellular debris and fibrin at the site of injury, they elaborate a host of mediators that induce fibroblast proliferation and ECM production. Mast cells and lymphocytes can contribute directly or indirectly to fibroblast proliferation and activation.
Deposition of ECM by these cells
As healing progresses, the number of proliferating fibroblast and new vessels decreases, however, the fibroblast progressively become more synthetic and hence there is increased deposition of ECM.Collagen synthesis in particular is critical to the development of strength in a healing wound site . collagen synthesis by fibroblasts begins early in wound healing and continues for several weeks, depending on the size of the wound.
The same growth factor that regulate fibroblast proliferation also participate in stimulating ECM synthesis.
As the scar matures there is progressive vascular regression , which eventually transforms the highly vascularized granulation tissue into a pale largely avascular scar.
Many growth factors are involved in the above processes including TGF-B , PDGF, and FGF as well as cytokines (IL- & TNF)
ECM and tissue remodeling
ECM continues to be modified and remodeled. The outcome of the repair process is in part a balance between ECM synthesis and degradation,the degradation of collagens and other ECM components is accomplished by a family of matrix metalloproteinase (MMP), which are dependent on zinc ion for their activity .MMPS include interstitial enzymes that degrade collagen, fibronectin, proteoglycans, synovial cells ) and their synthesis and secretion are regulated by growth factors, cytokines and other agents. Their synthesis may be suppressed pharmacologically with steroids.
Cutaneous wound healing
This is a process that involves both epithelial regeneration and the formation of connective tissue scar and is thus illustrative of the general principles that apply to wound healing in all tissues. The events are orchestrated by interplay of growth actors and ECM.
Cutaneous wound healing has three main phases:-
Inflammation
Formation of granulation tissue
ECM deposition and remodeling
Event in wound healing overlap to a great extent and cannot be completely separated from each other.
Based on the nature of the wound, the healing of the cutaneous wounds can occur by first or second intention.
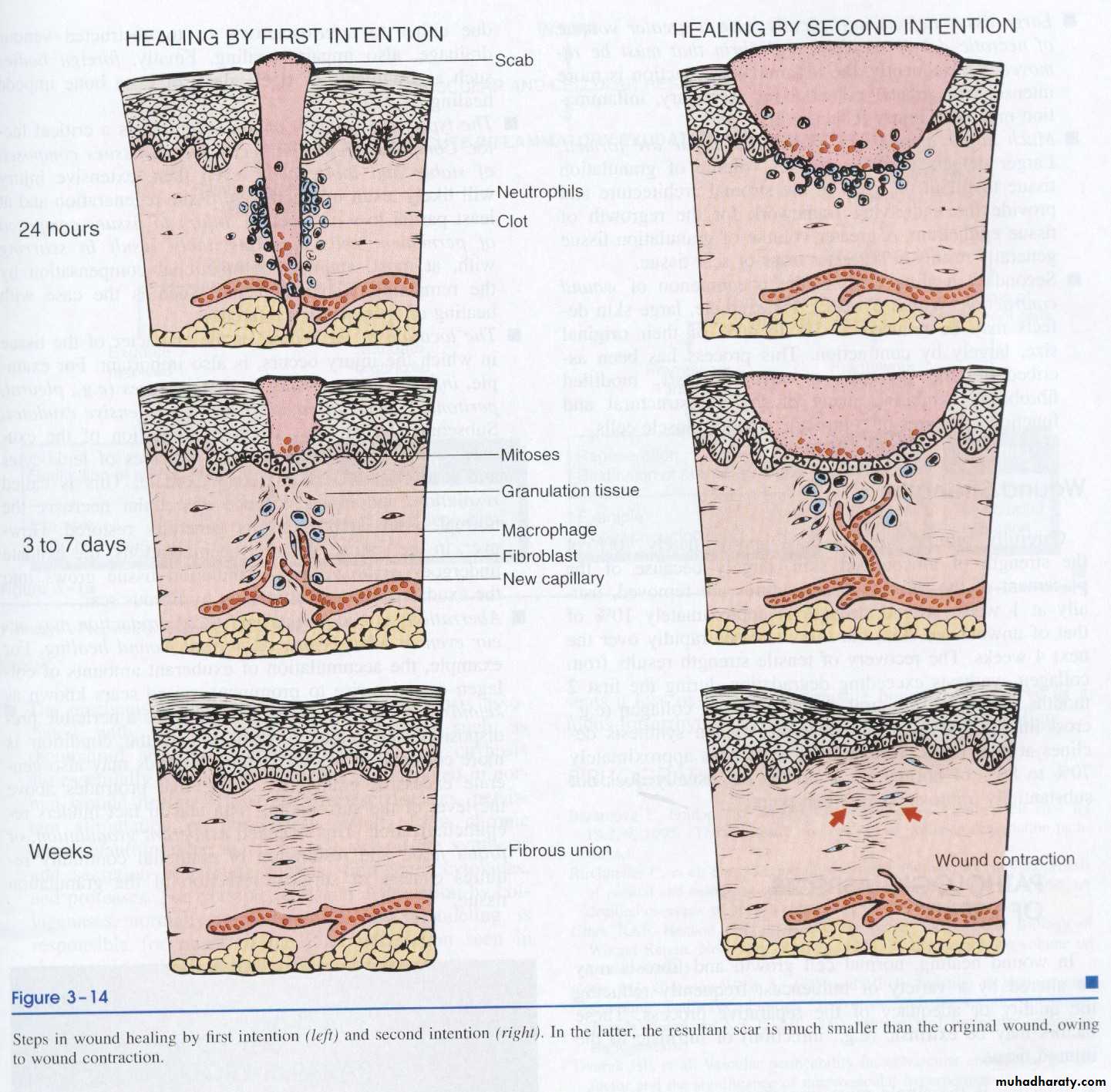
Healing by first intention :-
One of the simplest example of the wound repair is the healing of a clean uninfected surgical incision approximated by surgical sutures. This is referred to as primary union or healing by first intention.The incision causes only focal disruption of the epithelial basement membrane continuity and death of a relatively few epithelial and connective tissue cells. As a result, epithelial regeneration predominates over fibrosis. a small scar is formed but there is minimal wound contraction.
The narrow incisional space first fill with fibrin clotted blood.
Within 24 hr , neutrophils are seen at the incision margin migrating toward the fibrin clot.
Within 24 to 48 hrs, epithelial cells from both edges have begun to migrate and proliferate along the dermis. The cells meet at the midline, beneath the surface scab yielding a thin but continuous epithelial layer
Wound healing by primary and secondary union
By day 3 neutrophils have been largely replaced by macrophages and granulation tissue progressively invade the incision space. Epithelial cell proliferation continues yielding a thickened epidermal covering layer
By day 5, neovascularization reach its peak as granulation tissue fill the incisional space. The epidermis recover its normal thickness as differentiation of surface cells yields a mature epidermal architecture with surface keratinization
During the second week there is continued collagen accumulation and fibroblast proliferation that bridge the incision. The leukocytes infiltrate , edema and increase vascularity are diminished
The long process of blanching begins accomplished by increasing collagen deposition within the incisional scar and the regression of vascular channels.
By the end of the first month, the scar comprises acellular connective tissue largely devoid of inflammatory cells and covered by an essentially normal epidermis . the tensile strength of the wound increases with time . however the epidermal appendages destroyed in the line of the incision are permanently loss.
Arm, healing surgical incision. This image demonstrates healing by first intention which occur in clean, un infected wounds that have apposed edges.
Healing by second intention (healing by secondary union):-
• When cells or tissue loss is more extensive• the repair process is more complex,
• the inflammatory reaction is more intense,
• there is abundant development of granulation tissue and
• the wound contracts by the action of myofibroblast.
• This is followed by the accumulation of ECM and formation of a large scar.
• This mode of healing occurs in :
Large wound
Abscesses
Ulceration
After infarction in parenchymal organs
Wound with large tissue defect healed by secondary intention. Note the red granular appearance of the granulation tissue. A whitish- greenish- yellow neutrophilic exudates represent an inflammatory response to bacterial invasion of the wound.
KeloidKeloid nodular masses of of hyperplastic scar tissue, occur when the wound healing process runs unchecked. They are more commonly in people of African descent. surgical excision leads to repeated keloid formation.
Factors influencing wound healing
• Local• Type, size& location of wound.
• Adequacy of blood supply.
• Infection.
• Movement
• Presence of foreign body material.
• Exposure to ionizing radiation, & ultraviolet light.
B. Systemic
• Circulatory status.• Metabolic status.
• Systemic infection.
• Diabetes mellitus.
• Hormones.
• Hematological diseases; neutropnia.
• Renal failure.
• Tumor cachexia
Local factors
Type, size and location of the wound
• A clean, aseptic surgical wound heals faster than a wound produced by blunt trauma with abundant necrosis and irregular edges.
• Small wounds heal faster than large ones.
• Wounds that occur in richly vascularized areas such as the face heal faster than those that occur in poorly vascularized areas such as the foot.
Adequacy of blood supply
• Wound with poor blood supply heal slowly e.g. Leg wounds in patients with varicose veins.• Ischemia due to pressure produces bedsores. The same mechanism (pressure) prevents their healing. Ischemia due to arterial obstruction also prevents healing.
Infection
• Wounds provide a portal of entry for microorganisms. Infection delays or prevents healing.
Movement
• Early motion subjects the wound to persistent trauma.• Exercise also increases the circulating levels of glucocorticoids, which inhibit repair.
Exposure to ionizing radiation & ultraviolet light
• Irradiation of the wound blocks cell proliferation, retards granulation tissue formation & interferes with blood supply. The outcome is slow healing.
• Exposure of wounds to ultraviolet light accelerates the rate of healing.
Systemic factors
Circulatory status
• Cardiovascular status determines the blood supply to the injured area, which is important for wound healing. Poor healing attributed to old age is often due largely to impaired circulation.
Metabolic status
• Protein calorie malnutrition impairs healing.
• Vit. C deficiency (scurvy) impairs wound healing. This is because vit.c is involved in synthesis of collagen Thus in vit.c deficiency the collagen fibers show less cross-linking than normal & is more readily degraded.
• Zinc is required for collagen synthesis, so its deficiency will lead to delay wound healing.
Systemic infection
Leads to delay wound healing (multifactorial).Diabetes mellitus
Wounds in diabetics often become infected & in turn, infection makes the control of diabetes difficult, resulting in sever retardation or failure of healing.
Hormones
Corticosteroides impair wound healing due to
• inhibition of collagen synthesis,
• general depression of protein synthesis &
• anti-inflammatory effect, with scanty macrophage infiltrates & so lack of macrophage-derived growth factors.
Complications of wound healing
Wound dehiscence and Incisional hernias. A wound after a laparotomy may burst open. This occurs in 0.5-5% of all abdominal operations. Increased mechanical stress on the wound from vomiting, coughing, or ileus is a factor in over 90% of cases.
Incisional hernia is a late consequence of a weak abdominal scar.
Ulceration. Wounds ulcerate because of inadequate blood supply & vascularization. For example, leg wounds typically ulcerate.
Non-healing wounds develop in areas devoid of sensation, such as trophic or neuropathic ulcers, which may be seen in patients with spinal cord injury,
Excessive scar formation (Keloid). An excessive deposition of extracellular matrix at the wound site results in hypertrophic scar or keloid. Histologically, there are abundant, broad & irregular collagen bundles, with more capillaries and fibroblasts than would be expected for a scar of the same age. [] []
Excessive contraction (contracture). A decrease in wound size depends on the presence of myofibroblasts.