The pancreas
Dr. Ali Jaffer - Upper GI surgeon
Anatomy
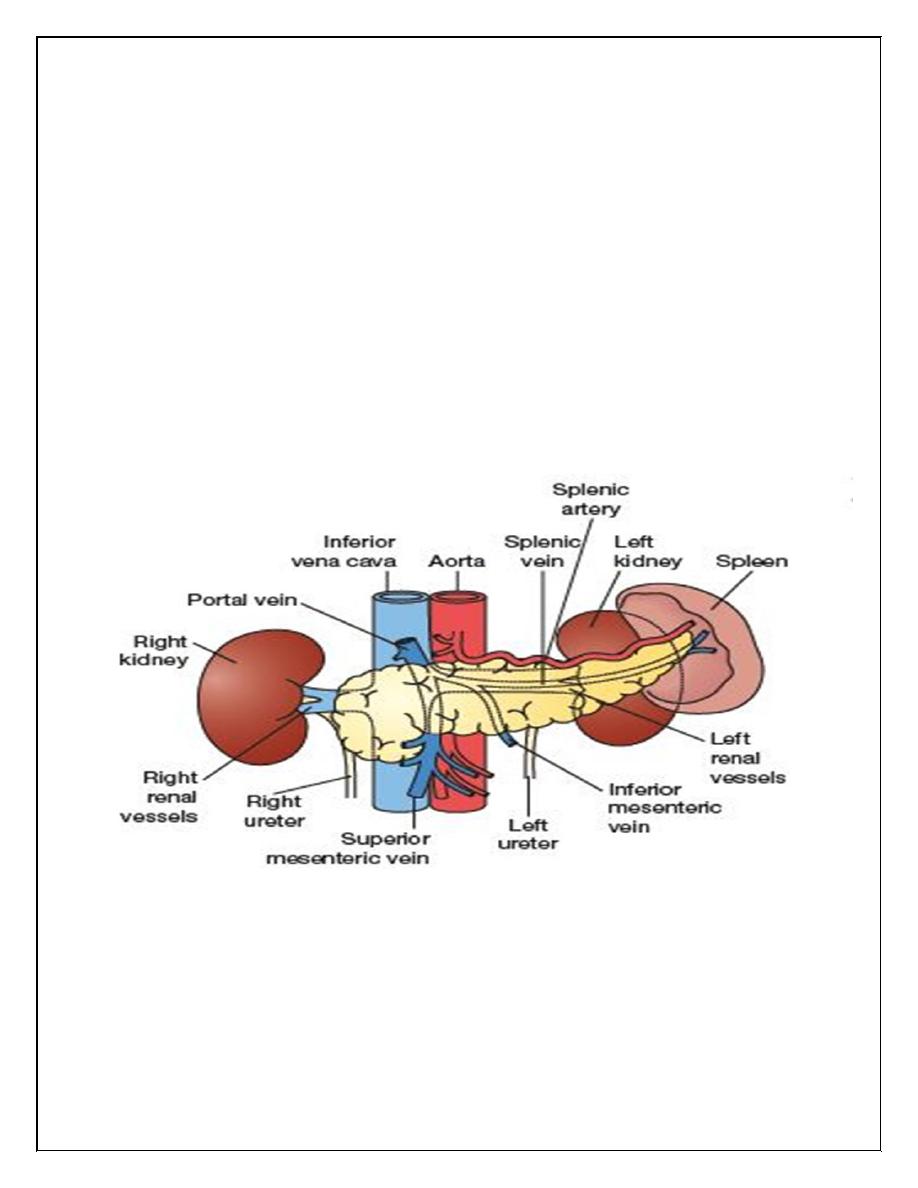
The pancreas is situated in the retroperitoneum. It is divided into a head, which occupies
30% of the gland by mass, and a body and tail, which together constitute 70%.
The head
lies within the curve of the duodenum, overlying the body of the second lumbar vertebra
and the vena cava.
The aorta and the superior mesenteric vessels lie behind the neck of the
gland.
Coming off the side of the pancreatic head and passing to the left and behind the
superior mesenteric vein is the uncinate process of the pancreas.
Behind the neck of the
pancreas, near its upper border, the superior mesenteric vein joins the splenic vein to form
the portal vein.
The tip of the pancreatic tail extends up to the splenic hilum.
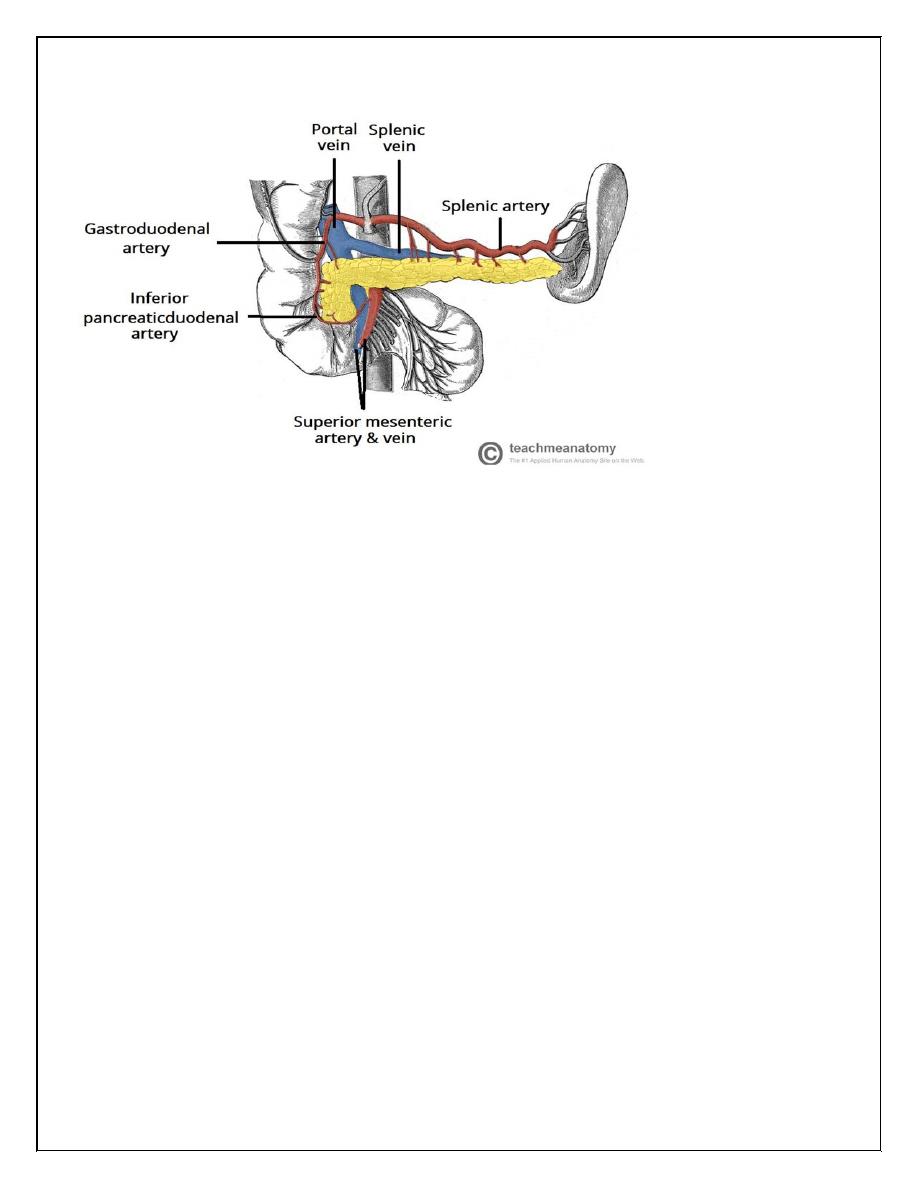
Vasculature
Arteries:
Splenic a., superior pancreaticoduodenal a
, inferior pancreaticoduodenal a.
Veins:
Splenic vein, superior mesenteric vein
Lympahatics:
The pancreas is drained by lymphatic
vessels that follow the arterial supply.
They empty into the pancreaticosplenal
nodes and the pyloric nodes, which in
turn drain
into the superior mesenteric and
coeliac lymph nodes.
INVESTIGATIONS
Estimation of pancreatic enzymes in body fluids
serum amylase, lipase
Pancreatic function tests
The nitroblue tetrazolium–para-aminobenzoic acid (NBT–PABA)
The pancreolauryl test
Faecal elastase
Imaging investigations
Ultrasonography: Ultrasonography is the initial investigation of choice in patients with
jaundice to determine whether or not the bile duct is dilated, the coexistence of gallstones or
gross disease within the liver such as metastases. It may also define the presence or absence of
a mass in the pancreas . However, obesity and overlying bowel gas often make interpretation
of the pancreas itself unsatisfactory.
b. Computed tomography: Most significant pathologies within the pancreas can be diagnosed
on high-quality CT scans.

c. Magnetic resonance imaging: Magnetic resonance cholangiography and pancreatography
(MRCP) may well replace diagnostic endoscopic cholangiography and pancreatography
(ERCP) as it is non-invasive and less expensive
d. Endoscopic retrograde cholangiopancreatography: ERCP is performed using a
side-viewing fibreoptic duodenoscope. The ampulla of Vater is intubated, and contrast is
injected into the biliary and pancreatic ducts to display the anatomy radiologically.
Diagnostic
and therapeutic
e. Endoscopic ultrasound: This is particularly useful in identifying small tumours that may
not show up well on CT or MRI, and in demonstrating the relationship of a pancreatic tumour
to major vessels nearby.
Transduodenal or transgastric fine needle aspiration (FNA) or Trucut
biopsy performed under EUS guidance avoids spillage of tumour cells into the peritoneal
cavity.
CONGENITAL ABNORMALITIES
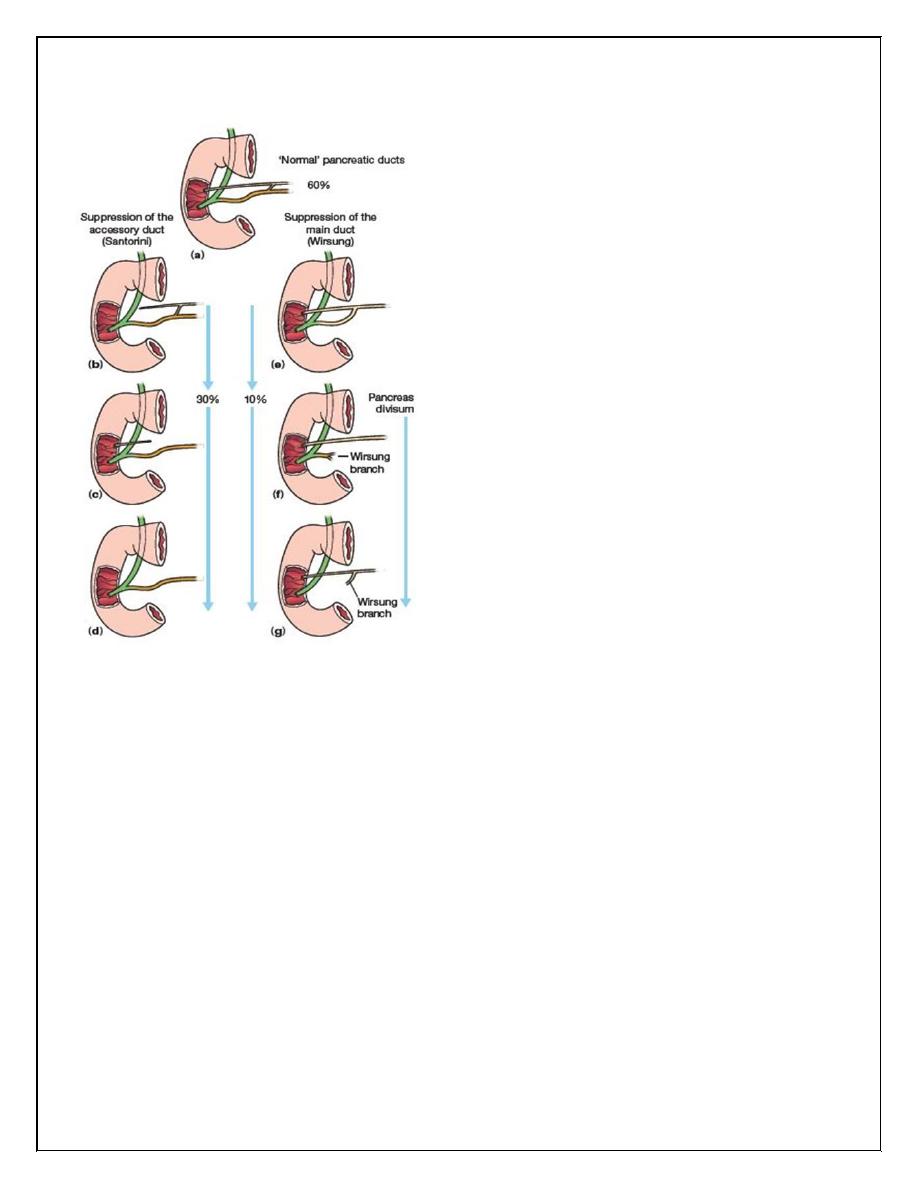
Pancreas divisum
occurs when the embryological ventral and dorsal parts of the pancreas fail to fuse. The dorsal
pancreatic duct becomes the main pancreatic duct and drains most of the pancreas through the
minor or accessory papilla. The incidence of pancreas divisum ranges from 5% to10%
.Pancreas divisum found incidentally in an asymptomatic person does not warrant any
intervention. The incidence of pancreas divisum ranges from 25–50% in patients with
recurrent acute pancreatitis, chronic pancreatitis and pancreatic pain. The minor papilla is
substantially smaller than the major papilla (and many of these patients probably have
papillary stenosis). A large volume of secretions flowing through a narrow papilla probably
leads to incomplete drainage, which may then cause obstructive pain or pancreatitis.
Idiopathic recurrent pancreatitis, pancreas divisum should be excluded. The diagnosis can be
arrived at by MRCP, EUS or ERCP, augmented by injection of secretin if necessary.
Endoscopic sphincterotomy and stenting of the minor papilla may relieve the symptoms.
Surgical intervention can take the form of sphincteroplasty, pancreatojejunostomy or even
resection of the pancreatic head.
Annular pancreas
This is the result of failure of complete rotation of the ventral pancreatic bud during
development, so that a ring of pancreatic tissue surrounds the second or third part of the
duodenum.
It is most often seen in association with congenital duodenal stenosis or atresia
and is therefore more prevalent in children with Down syndrome. The usual treatment is
bypass (duodenoduodenostomy). The disease may occur in later life as one of the causes of
pancreatitis.
Ectopic pancreas
Islands of ectopic pancreatic tissue can be found in the submucosa in parts of the stomach,
duodenum or small intestine (including Meckel’s diverticulum), the gall bladder, adjoining the
pancreas, in the hilum of the spleen and within the liver.
Ectopic pancreas may also be found
in the wall of an alimentary tract duplication cyst.
INJURIES TO THE PANCREAS

External injury
Presentation and management
The pancreas, its somewhat protected location in the retroperitoneum, is not frequently
damaged in blunt abdominal trauma. If there is damage to the pancreas, it is often concomitant
with injuries to other viscera, especially the liver, the spleen and the duodenum. Pancreatic
injuries may range from a contusion or laceration of the parenchyma without duct disruption
to major parenchymal destruction with duct disruption (sometimes complete transection) and,
rarely, massive destruction of the pancreatic head.
Blunt pancreatic trauma usually presents with epigastric Pain with the progressive
development of more severe pain due to the sequelae of leakage of pancreatic fluid into the
surrounding tissues. A rise in serum amylase occurs in most cases.
A CT scan of the pancreas
will delineate the damage that has occurred to the pancreas.
If there is doubt about duct
disruption, an urgent ERCP should be sought. MRCP may also provide the answer.
Support with intravenous fluids and a ‘nil by mouth’ regimen should be instituted while these
investigations are performed.
It is preferable to manage conservatively at first, investigate
and, once the extent of the damage has been ascertained, undertake appropriate action.
Operation is indicated if there is disruption of the main pancreatic duct; in almost all other
cases, the patient will recover with conservative management.
Penetrating trauma to the upper abdomen or the back carries a higher chance of pancreatic
injury.
In penetrating injuries, especially if other organs are injured and the patient’s condition
is unstable, there is a greater need to perform an urgent surgical exploration.
Sequlae
Death, in acute phase as a result of bleeding from associated injuries.
Persistent drain output in 1/3 of patients
Duct stricture resulting in recurrent episodes of pancreatitis.
Pancreatic pseudocyst.
Iatrogenic injury
This can occur in several ways:
● Injury to the tail of the pancreas during splenectomy, resulting in a pancreatic fistula.

● Injury to the pancreatic head and the accessory pancreatic duct (Santorini), which is the
main duct in 7% of patients, during Billroth II gastrectomy
● Enucleation of islet cell tumours of the pancreas can result in fistulae.
● Duodenal or ampullary bleeding following sphincterotomy.
Pancreatic fistula
Management of pancreatic fistulae
Tests
● Measure amylase level in fluid
● Determine the anatomy of the fistula
● Check if the main pancreatic duct is blocked or disrupted
Measures
● Correct fluid and electrolyte imbalances
● Protect the skin
● Drain adequately
● Parenteral or nasojejunal feeding
● Octreotide to suppress secretion
● Relieve pancreatic duct obstruction if possible (ERCP andstent)
● Treat underlying cause
Acute pancreatitis
The underlying mechanism of injury in pancreatitis is thought to be premature activation of
pancreatic enzymes within the pancreas, leading to a process of autodigestion.
Once cellular
injury has been initiated, the inflammatory process can lead to pancreatic oedema,
haemorrhage and, eventually, necrosis.
As inflammatory mediators are released into the
circulation, systemic complications can arise, such as haemodynamic instability, bacteraemia
(due to translocation of gut flora), acute respiratory distress syndrome and pleural effusions,
gastrointestinal haemorrhage, renal failure and disseminated intravascular coagulation
(DIC).
The majority of patients will have a mild attack of pancreatitis, the mortality from
which is around 1%. Severe acute pancreatitis is seen in 5–10% of patients, and is
characterised by pancreatic necrosis, a severe systemic inflammatory response and often
multi-organ failure.
In those who have a severe attack of pancreatitis, the mortality varies
from 20 to 50%.

Acute pancreatitis has an early phase that usually lasts a week. It is characterised by a
systemic inflammatory response syndrome (SIRS) which – if severe – can lead to transient
or persistent organ failure (deemed persistent if it lasts for over 48 hours). About one-third of
deaths occur in the early phase of the attack, from multiple organ failure. The late phase is
seen typically in those who suffer a severe attack, and can run from weeks to months. It is
characterised by persistent systemic signs of inflammation, and/or local complications,
particularly fluid collections and peripancreatic sepsis. Deaths occurring after the first week
of onset are often due to septic complications.
Possible causes of acute pancreatitis
● Gallstones
● Alcoholism ( Gallstones and Alcoholism contribute to 50-70% of cases)
● Post ERCP 1-3 %
● Abdominal trauma
● Following biliary, upper gastrointestinal or cardiothoracic surgery
● Ampullary tumour
● Drugs (corticosteroids, azathioprine, asparaginase, valproic acid, thiazides, oestrogens)
● Hyperparathyroidism
● Hypercalcaemia
● Pancreas divisum
● Autoimmune pancreatitis
● Hereditary pancreatitis
● Viral infections (mumps, coxsackie B)
● Malnutrition
● Scorpion bite
● Idiopathic
Clinical presentation
Pain characteristically develops quickly, reaching maximum intensity within minutes and
persists for hours or even days. The pain is frequently severe, constant and refractory to the
usual doses of analgesics. epigastrium but may be localised to either upper quadrant or felt
diffusely throughout the abdomen. There is radiation to the back in about 50% of patients,
and some patients may gain relief by sitting or leaning forwards.
Nausea, repeated vomiting
and retching. The retching may persist despite the stomach being kept empty by nasogastric
aspiration. Hiccoughs may be due to gastric distension or irritation of the diaphragm.
On examination, the patient may be well or gravely ill.
Tachypnoea , tachycardia is usual,
and hypotension .
The body temperature is often normal or even subnormal. Mild icterus can
be caused by biliary obstruction in gallstone pancreatitis.
Bleeding into the fascial planes can

produce bluish discolouration of the flanks (Grey Turner’s sign) or umbilicus (Cullen’s sign).
Neither sign is pathognomonic of acute pancreatitis; Cullen’s sign was first described in
association with rupture of an ectopic pregnancy. Subcutaneous fat necrosis may produce
small, red, tender nodules on the skin of the legs.
Abdominal examination may reveal
distension due to ileus. There is usually muscle guarding in the upper abdomen.
A pleural
effusion is present in 10–20% of patients. Pulmonary oedema and pneumonitis
Investigations
A serum amylase level three times above normal
serum levels lipase
contrast- enhanced CT.
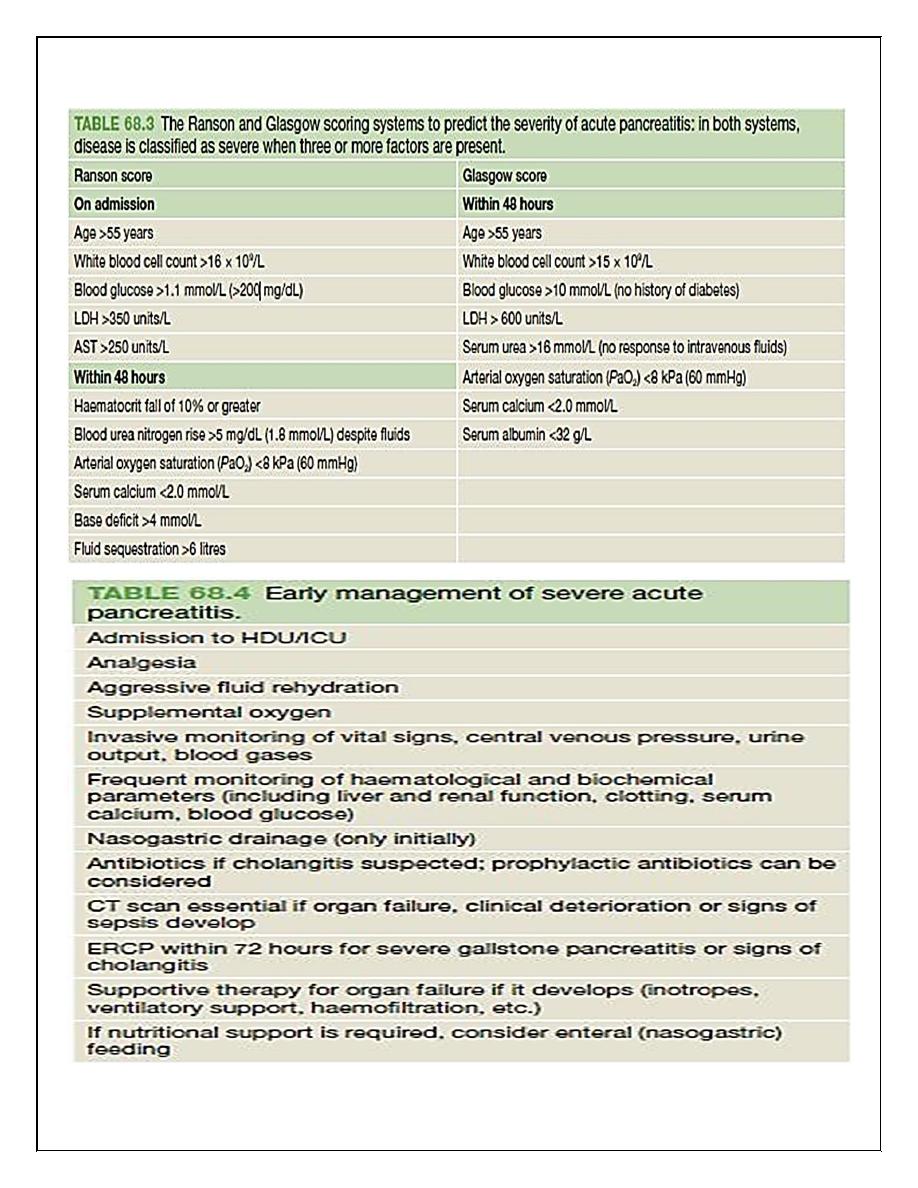
ASSESSMENT OF SEVERITY
Severity stratification assessments should be performed in patients at 24 hours, 48 hours and
7 days after admission.
The Ranson and Glasgow scoring systems are specific for acute
pancreatitis, and a score of 3 or more at 48 hours indicates a severe attack
Regardless of the system used, persisting organ failure indicates a severe attack. A serum
C-reactive protein level >150 mg/L at 48 hours after the onset of symptoms is also an
indicator of severity.
Mild acute pancreatitis:
no organ failure;
no local or systemic complications.
Moderately severe acute pancreatitis:
organ failure that resolves within 48 hours (transient
organ failure); and/or
local or systemic complications without persistent organ failure.
Severe acute pancreatitis:
persistent organ failure (>48 hours);
single organ failure;
multiple organ failure.

PSEUDOCYST
A pseudocyst is a collection of amylase-rich fluid enclosed in a well-defined wall of fibrous
or granulation tissue. Formation of a pseudocyst requires 4 weeks or more from the onset of
acute pancreatitis.
A pseudocyst is usually identified on ultrasound or a CT scan.
EUS and aspiration of the cyst
fluid. The fluid should be sent for measurement of carcinoembryonic antigen (CEA) levels,
amylase levels and cytology.
Fluid from a pseudocyst typically has a low CEA level, and
levels above 400 ng/mL are suggestive of a mucinous neoplasm.
Pseudocyst fluid usually
has a high amylase level, but that is not diagnostic, as a tumour that communicates with the
duct system may yield similar findings. Cytology typically reveals inflammatory cells in
pseudocyst fluid.
Pseudocysts will resolve spontaneously in most instances,
but complications can develop .
Pseudocysts that are thick-walled or large (over 6 cm in diameter), have lasted for a long
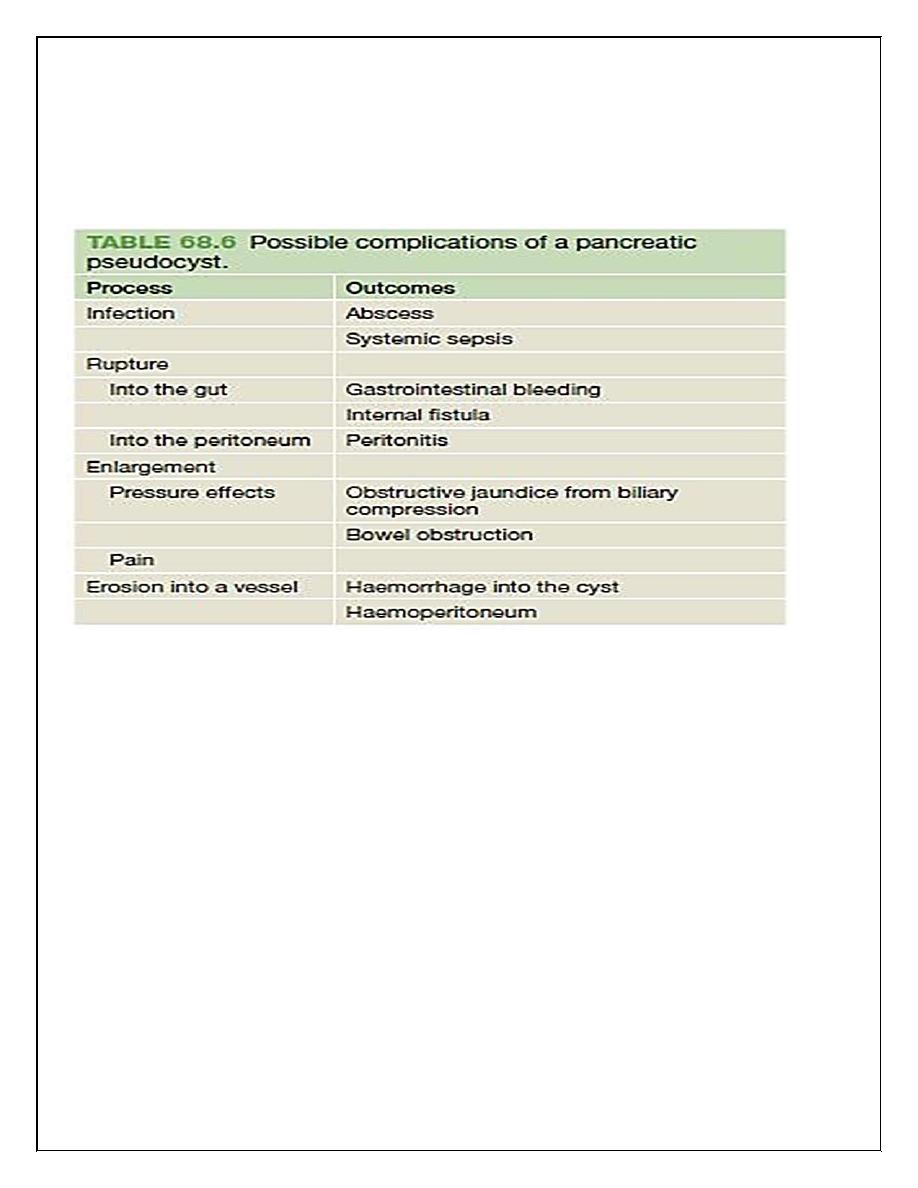
time (over 12 weeks), or have arisen in the context of chronic pancreatitis are less likely to
resolve spontaneously.
Therapeutic interventions are advised only if the pseudocyst causes
symptoms, if complications develop, or if a distinction has to be made between a pseudocyst
and a tumour.
Percutaneous drainage to the exterior under radiological guidance should be avoided. It
carries a very high likelihood of recurrence. Moreover, it is not advisable unless one is
absolutely certain that the cyst is not neoplastic and that it has no communication with the
pancreatic duct (or else a pancreaticocutaneous fistula will develop).
Endoscopic drainage
usually involves puncture of the cyst through the stomach or duodenal wall under EUS
guidance, and placement of a tube drain with one end in the cyst cavity and the other end in
the gastric lumen.
Surgical drainage involves internally draining the cyst into the gastric or
jejunal lumen
STERILE AND INFECTED PANCREATIC NECROSIS
Refers to a diffuse or focal area of non-viable parenchyma.
Acute necrotic collection (ANC). This is typically an intra or extrapancreatic collection
containing fluid and necrotic material, with no definable wall.
Gradually, over a period of
over 4 weeks, this may develop a well-defined inflammatory capsule, and evolve into what
is termed walled-off necrosis (WON).

Collections associated with necrotising pancreatitis are sterile to begin with but often
become subsequently infected, probably due to translocation of gut bacteria.
Infected
necrosis is associated with a mortality rate of up to 50%.
Sterile necrotic material should not
be drained or interfered with. But if the patient shows signs of sepsis, then one should
determine whether the collection is infected
Dx: aspiration under Ct scan guidance. (the needle should never pass through hollow
viscera). Rx: if the aspirated is purulent, the patient should be kept on antibiotics according
to sensitivity and drain inserted for drainage. The drain should be with larger size when the
aspirate is thick content. if sepsis persist this need pancreatic necrosectomy. Patients with
peripancreatic sepsis are ill for long periods of time, and may require management in an
intensive care unit. Nutritional support is essential. The parenteral and nasojejunal
approaches are more popular (on the assumption that they rest the pancreas).
Chronic pancreatitis
Chronic pancreatitis is a progressive inflammatory disease in which there is irreversible
destruction of pancreatic tissue. Its clinical course is characterised by severe pain and, in the
later stages, exocrine and endocrine pancreatic insufficiency.
Aetiology and pathology
Alcoholism 60-70%
Duct obstruction due to trauma, acute pancreatitis or pancreatic cancer
Congenital anomalies Hereditary pancreatitis
Cystic fibrosis
Hyperlipidemia
Hypercalcemia
Autoimmune pancreatistis (IgG4 phenomena)
Idioathic
*** At the onset of the disease, the pancreas may appear normal. Later, the pancreas enlarges
and becomes hard as a result of fibrosis. The ducts become distorted and dilated with areas
of both stricture formation and ectasia. Calcified stones form within the ducts. The ducts
may become occluded with a gelatinous proteinaceous fluid and debris, and inflammatory
cysts may form. Histologically, the lesions affect the lobules, producing ductular metaplasia
and atrophy of acini, hyperplasia of duct epithelium and interlobular fibrosis.
Clinical features
Pain; the site of the pain is depend on the site of the disease. Head of pancreas; the pain felt
in the epigastric and right subcostal region, whereas if the disease is limited to the body and
tail, the pain felt in the left subcostal region and back.

Nausea is common, vomiting may develop.
Weight loss
The patient’s lifestyle is gradually destroyed by pain, analgesic dependence, weight loss and
inability to work.
Loss of exocrine function leads to steatorrhoea in more than 30% of patients with chronic
pancreatitis.
Loss of endocrine function and the development of diabetes
Infection
Investigations
Only in the early stages of the disease will there be a rise in serum amylase.
Tests of pancreatic function merely confirm the presence of pancreatic insufficiency or that
more than 70% of the gland has been destroyed.
Pancreatic calcifications may be seen on abdominal X-ray. CT or MRI scan will show the
outline of the gland, the main area of damage and the possibilities for surgical correction, An
MRCP will identify the presence of biliary obstruction and the state of the pancreatic duct.
ERCP is the most accurate way of elucidating the anatomy of the duct.
EUS
Medical treatment of chronic pancreatitis
Treat the addiction, Help the patient to stop alcohol consumption and tobacco smoking,
Involve a dependency counsellor or a psychologis, Alleviate abdominal pain
Eliminate obstructive factors (duodenum, bile duct, pancreatic duct)
Escalate analgesia in a stepwise fashion, Refer to a pain management specialist, For
intractable pain, consider CT/EUS-guided coeliac axis block
Nutritional and pharmacological measures, Diet: low in fat and high in protein and
carbohydrates, Pancreatic enzyme supplementation with meals
Correct malabsorption of the fat-soluble vitamins and vitaminB12, Micronutrient therapy
with methionine, vitamins C & E, selenium (may reduce pain and slow disease progression)
Steroids (only in autoimmune pancreatitis, for relief of symptoms)
Medium-chain triglycerides in patients with severe fat malabsorption (they are directly
absorbed by the small intestine without the need for digestion)
Reducing gastric secretions may help

Treat diabetes mellitus
Endoscopic, radiological or surgical interventions are indicated mainly to relieve obstruction
of the pancreatic duct, bile duct or the duodenum, or in dealing with complications (e.g.
pseudocyst, abscess, fistula, ascites or variceal haemorrhage).
Prognosis
Chronic pancreatitis is a difficult condition to manage. Patients often suffer a gradual decline
in their professional, social and personal lives.
The pain may abate after a surgical or percutaneous intervention, but tends to return over a
period of time.
In a proportion of patients, the inflammation may gradually burn out over a
period of years, with disappearance of the pain, leaving only the exocrine and endocrine
insufficiencies.
Development of pancreatic cancer is a risk in those who have had the disease for more than
20 years.
New symptoms or a change in the pattern of symptoms should be investigated and
malignancy excluded.
