
Anti-Diabetic Drugs
Bassim I Mohammad
Specialist Physician-Assistant Professor
College of Pharmacy/ Al Qadisiyah University
Iraq

DM is an elevated blood glucose level associated with absent ,
or inadequate pancreatic insulin secretion, with or without
concurrent impairment insulin action
Diabetes Mellitus (Definition)

TYPE 1
: characterized by
beta cell destruction
and severe or
absolute insulin deficiency (immune form)
TYPE 2
: characterized by
tissue resistance to the action of insulin
combined with a relative deficiency in insulin secretion
TYPE 3
: refers to multiple other specific causes of an elevated
blood glucose: pancreatectomy, pancreatitis, drug therapy…etc)
TYPE 4
:Gestational diabetes is defined as any abnormality in
glucose levels noted for the first time
during pregnancy
Diabetes Mellitus (Classification)

Diabetes Mellitus (Clinical Features)
Polyuria
Polydipsia
Polyphagia
Weight loss

Criteria for the Diagnosis of Diabetes
A1C ≥6.5%
OR
Fasting plasma glucose (FPG)
≥126 mg/dL (7.0 mmol/L)
OR
2-h plasma glucose ≥200 mg/dL
(11.1 mmol/L) during an OGTT
OR
A random plasma glucose ≥200 mg/dL
(11.1 mmol/L)
ADA. 2. Classification and Diagnosis. Diabetes Care 2015;38(suppl 1):S9; Table 2.1

FPG 100–125 mg/dL (5.6–6.9 mmol/L): IFG
OR
2-h plasma glucose in the 75-g OGTT
140–199 mg/dL (7.8–11.0 mmol/L): IGT
OR
A1C 5.7–6.4%
*For all three tests, risk is continuous, extending below the lower limit of a range and becoming
disproportionately greater at higher ends of the range.
ADA. 2. Classification and Diagnosis. Diabetes Care 2015;38(suppl 1):S10; Table 2.3
Categories of Increased Risk for Diabetes
(Prediabetes)*
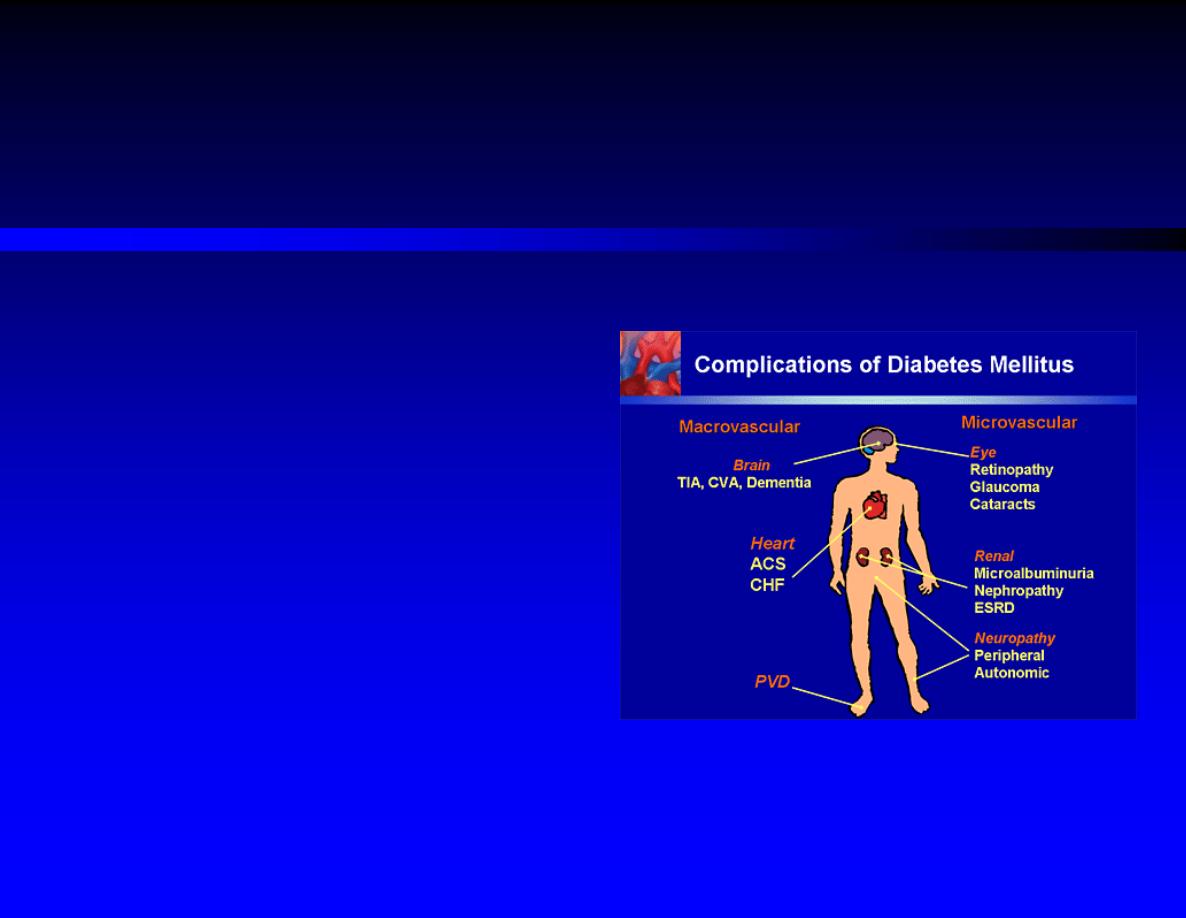
DM (Complications)
1)
Acute Complications:
Diabetic ketoacidosis
Nonketotic hyperosmolar coma
2)
Chronic Complications:
(micro/macrovascular)

Type 1: Insulin is a must, education and nutrition control
Type 2: education, Food control, exercise, OAD
(1) Increase
insulin secretion
;
(2) Increase the
sensitivity
of target organs to insulin;
(3) Decrease
glucose absorption
(4) Insulin needed serious complications or an emergency
Gestational Diabetes (Insulin)
Diabetes Mellitus (Treatment Strategies)

Insulin
Oral Anti-Diabetic Agents
Diabetes Mellitus (Treatment)

Chemistry , Physiology
Pharmacological Effect
Mechanism of action
Clinical Uses
Types and Preparations and mixtures
Delivery systems
Complications
Insulin

Insulin is a small protein contains 51 amino acids arranged in
two chains (A and B) linked by disulfide bridges.
Proinsulin in the Golgi apparatus of beta cells, packaged into
granules, and hydrolyzed into
insulin and C-peptide
by removal
of four amino acids
Insulin and C-peptide (has no physiological action) are secreted
in equimolar amounts in response to all insulin secretagogues.
Insulin (Chemistry and Physiology)

Insulin is released from pancreatic beta cells at a
low basal
rate and at a
much higher stimulated
rate in response to a
variety of stimuli, especially glucose.
Oral glucose
elicits more insulin secretion than dose IV
glucose; because
oral administration of glucose elicits gut
hormones which augment the insulin response
Insulin (Chemistry and Physiology)

Insulin release Stimulators/ Inhibitors
Stimulators
Inhibitors
Glucose (most important)
α-Adrenergic stimulation inhibits release
(most important)
β-Adrenergic stimulation
Glucagon
Amino acid
Somatostation
Cholecystokinin
Leptin
Elevated intracellular Ca2+
Hypoxia
Drugs (eg sulfonylureas)
Drugs ( eg e diazoxide, colchicine)

The liver and kidney are the two main organs that remove
insulin from the circulation
Endogenous insulin: 60% liver, 40% kidney
Exogenous insulin: 60% kidney,40% liver
The half-life of circulating insulin (endogenous) is 3–5
minutes
Is not teratogenic
Insulin (Degradation)

Carbohydrate metabolism
: reducing blood glucose levels by
glycogenolysis ↓, glycogen synthesis ↑, gluconeogenesis ↓
(ketone badies )
Lipid metabolism
: fat synthesis ↑, lipolysis↓ , plasma FFA ↓
Protein metabolism
: active transport of aa ↑, incorporation of
amino acids into protein ↑, protein catabolism ↓
HR↑ , myocardial contractility, renal blood flow ↓
Potassium
: k+ uptake into cells ↑
Insulin (Pharmacological effects)
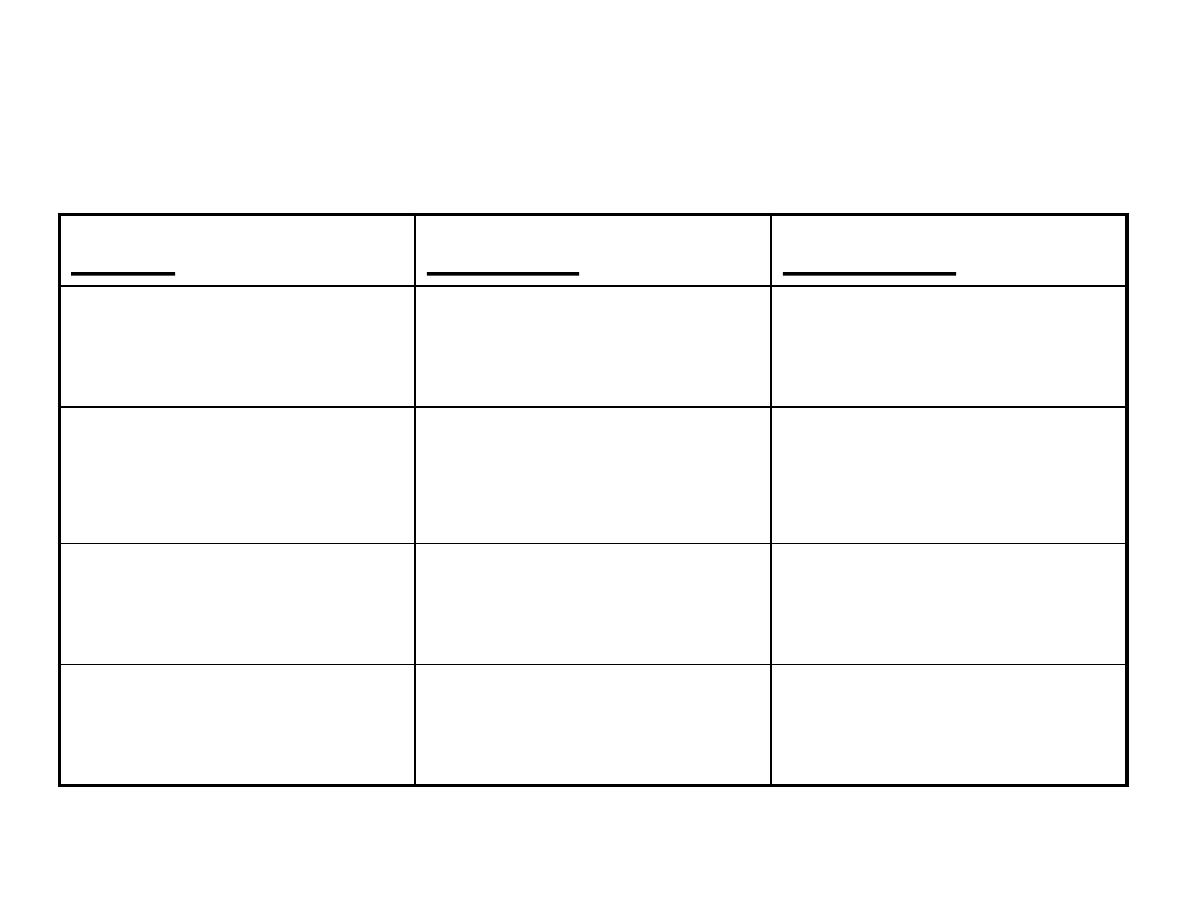
Action of Insulin on Various Tissues
Liver
Muscle
Adipose
↓ glucose production ↑ Glucose transport
↑ glucose transport
↑ glycolysis
↑ glycolysis
↑ lipogenesis&
lipoprotein lipase
activity
↑ TG synthesis
↑ glycogen deposition ↓ intracellular
lipolysis
↑ Protein synthesis
↑ protein synthesis

Insulin binds to specialized receptors (found on the
membranes of most tissues particularly target tissues, ie, liver,
muscle, and adipose tissue) with
high specificity and affinity
.
Insulin receptor consists of two subnits. An
α subunit
, which
constitutes the
recognition site
and
β subunits
, which contains
a tyrosine kinase
.
Insulin (Mechanism of action)

The binding of an insulin molecule to the α subunits activates
the receptor and through a conformational change, facilitates
mutual
phosphorylation
of tyrosine residues on the β subunits
and tyrosine kinase activity.
These process results in multiple effects, including increase in
glucose uptake (
translocation of GLUT 4
), increased glycogen
synthase activity and increased glycogen formation; multiple
effects on protein synthesis, lipolysis, and lipogenesis ..etc
Insulin (Mechanism of action)
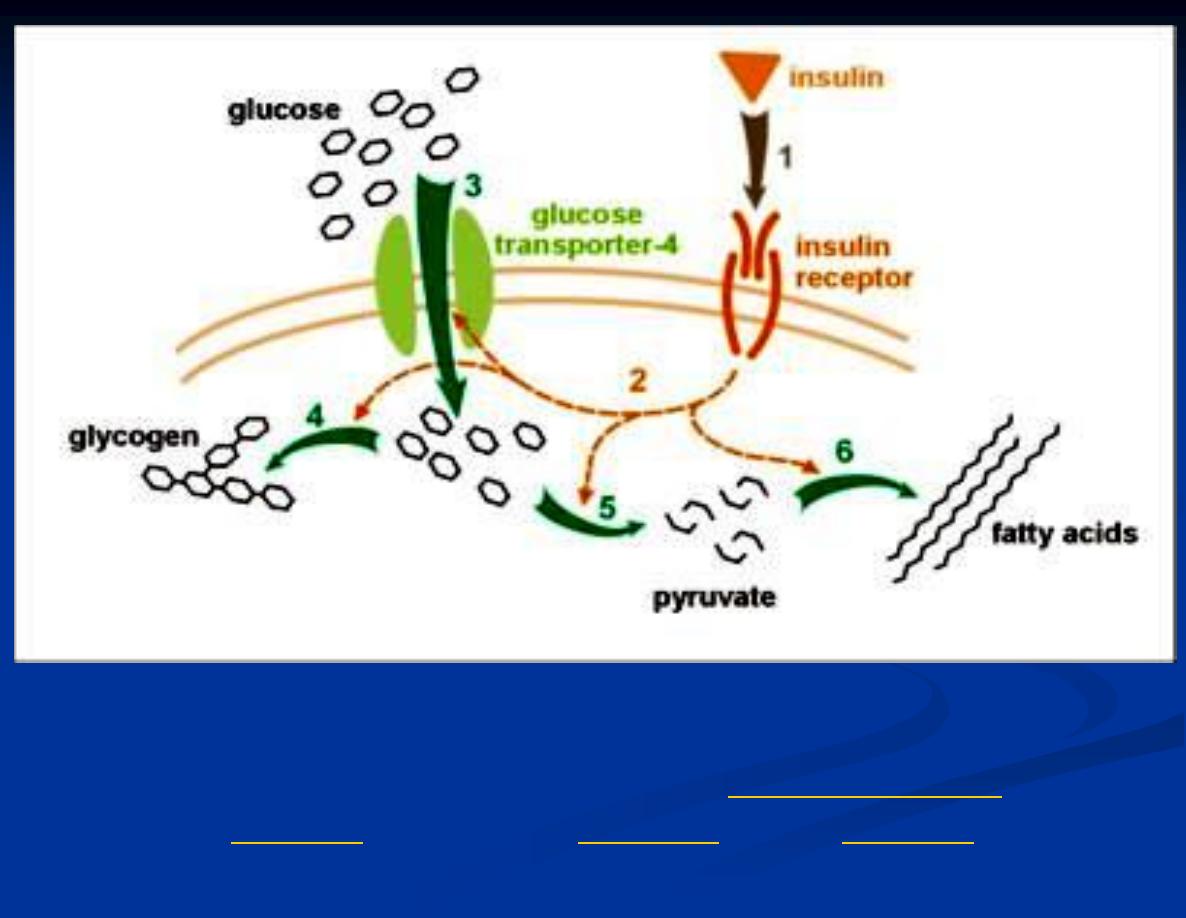
19
Effect of insulin on glucose uptake and metabolism. Insulin binds to its
receptor (1) which in turn starts many protein activation cascades (2). These
include: translocation of Glut-4 transporter to the
of glucose (3),
synthesis (6).

Insulin-dependent patients with diabetes mellitus (type 1 diabetes mellitus)
Insulin-independent patients: failure to other drugs
Diabetic complications: diabetic, hyperosmotic nonketotic coma
Critical (stress) situations of diabetic patients: fever, severe infection,
pregnancy, trauma, operation
Others: promotion of K+ uptake into the cells, pshychiatric disorders
Insulin (Clinical uses)

Commercial insulin preparations differ in a number of ways,
recombinant DNA production techniques, amino acid
sequence, concentration, solubility, and the time of onset and
duration of their biologic action
Insulin (Available Preparations)

Insulin (4 Injectable Available Preparations)
1. Rapid (Ultra-short) acting (very fast onset and short duration)
2. Short-acting (Regular) (rapid onset of action)
3. Intermediate-acting
4. Long-acting (slow onset of action)

Rapid acting and short-acting
insulins are dispensed as
clear solutions
at
neutral pH and contain small amounts of zinc to improve their stability
and shelf life
Intermediate-acting NPH insulin have been modified to provide
prolonged action and are dispensed as a
turbid suspension
at neutral pH
with protamine in phosphate buffer (neutral protamine Hagedorn [NPH]
insulin)
Insulin glargine and insulin detemir
are clear, soluble
long-acting insulins
Insulin (Available Preparations)

Three rapid-acting Insulin analogs Are Commercially Available (
Insulin
lispro, Insulin aspart, Insulin glulisine)
Permit more physiologic prandial insulin replacement because their
rapid
onset and early peak action more
Their duration of action is 4–5 hours, which decreases the risk of late
postmeal hypoglycemia
Have the
lowest variability of absorption
(approximately 5%) of all
available commercial insulins
Rapid-acting Insulin

Its effect appears within 30 minutes, peaks between 2-3 hours after
subcutaneous injection, and generally lasts 5–8 hours
The hexameric nature of regular insulin causes a
delayed onset and
prolongs the time to peak action
After S/C injection, the insulin hexamersare too large and bulky to be
transported across the vascular endothelium into the bloodstream. As the
hexamers break down into dimers and finally monomers. This results in
three rates of absorption of the injected insulin
, with the final monomeric
phase having the fastest uptake out of the injection
Short-acting (Regular) Insulin

Clinically, when regular insulin is administered at mealtime, the blood
glucose rises faster than the insulin with resultant early postprandial
hyperglycemia and an increased risk of late postprandial hypoglycemia
.
Therefore, regular insulin should be injected 30–45 or more minutes
before the meal to minimize the mismatching
Short-acting (Regular) Insulin

The delayed absorption, dose-dependent duration of action, and variability
of absorption (
∼ 25%)
of regular human insulin frequently results in a
mismatching of insulin availability with need, and its use is declining
It is particularly useful for IV therapy in the management of DKA and
when the insulin requirement is changing rapidly, such as after surgery or
during acute infections.
Limitations of regular insulin, namely, highly dose dependent PKs and
PDs profiles, and variability in absorption
Short-acting (Regular) Insulin

NPH insulin is an intermediate-acting insulin is a suspension of
crystalline zinc insulin combined with protamine (a polypeptide).
The conjugation with protamine
delays its onset of action and prolongs it
effectiveness.
Has an onset of approximately 2–5 hours and duration of 4–12 hours and
is usually mixed with regular, lispro, aspart, or glulisine insulin
The action of NPH is highly unpredictable, and its variability of
absorption is over 50%
Intermediate-acting NPH (neutral protamine Hagedorn, or
isophane) Insulin

Insulin glargine is a soluble, “peakless” (ie, having a broad plasma
concentration plateau), long-acting insulin analog
Achieves a maximum effect after 4–6 hours. This maximum activity is
maintained for 11–24 hours or longer. Glargine is usually given once daily
Should not be mixed with other insulins
(separate syringes must be used)
Long-acting Insulin (Glargine)
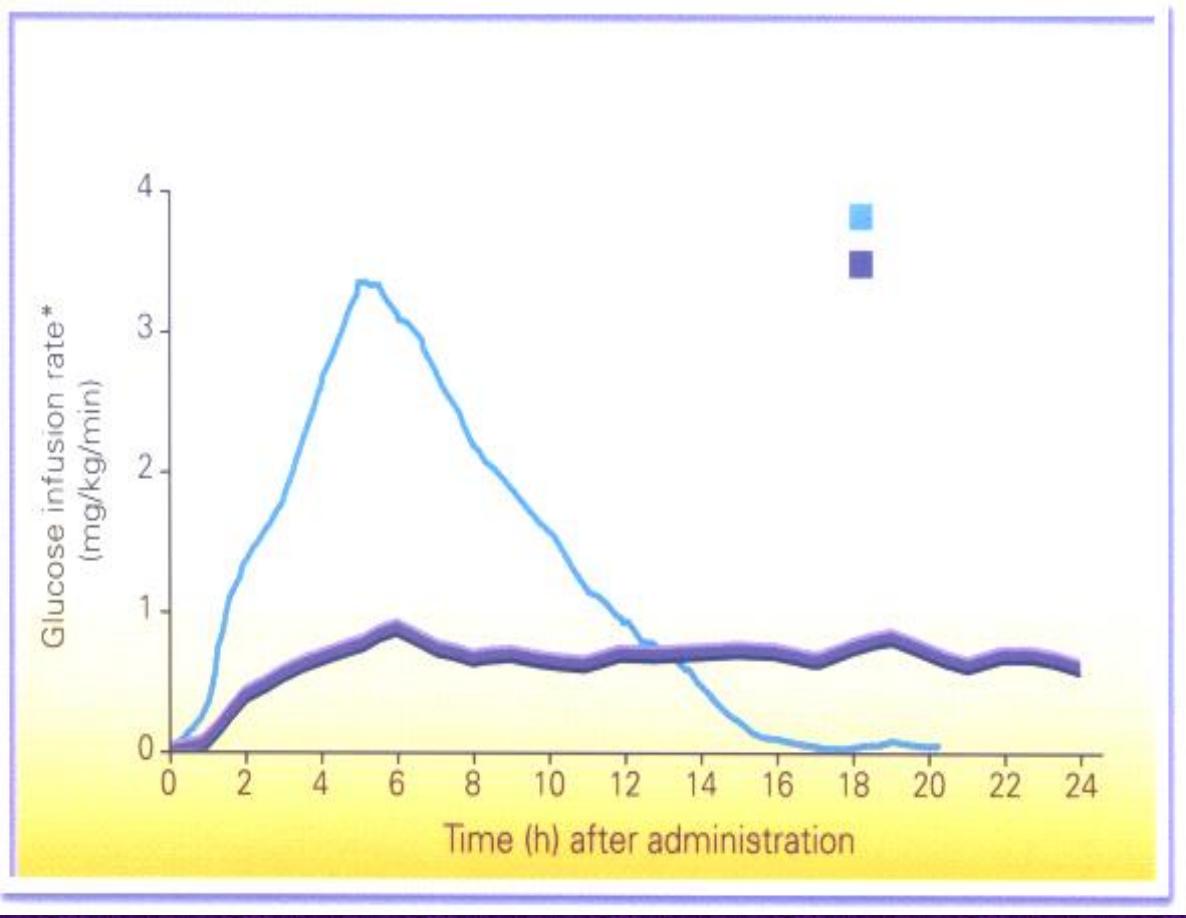
30
Profile of Insulin Glargine vs NPH
Glargine
NPH

This insulin is the most recently developed long-acting insulin analog
Has the most reproducible effect of the intermediate- and long-acting
insulins, and its use is associated with
less hypoglycemia
than NPH
insulin
Has a dose-dependent onset of action of 1–2 hours and duration of action
of more than 12 hours.
It is given twice daily to obtain a smooth background insulin level.
Long-acting Insulin (Insulin detemir)

Because NPH insulins require several hours to reach adequate therapeutic
levels, their use in diabetic patients usually requires supplements of rapid- or
short-acting insulin before meals.
These are often mixed together in the same syringe before injection. Insulin
lispro, aspart, and glulisine can be
acutely mixed
(ie, just before injection)
with NPH insulin without affecting their rapid absorption.
Premixed formulations of 70%/30% NPH/regular continue to be available.
These preparations have all the limitations of regular insulin, namely, highly
dose dependent PKs and PDs profiles, and variability in absorption
Mixtures (combinations) Of Insulins

Standard Delivery (conventional disposable syringes)
Portable Pen Injectors
Continuous Subcutaneous Insulin Infusion Devices ( Insulin Pumps)
Inhaled Insulin (dry powder formulation) used in adult, peak level reached
15 minutes and last for 3 hours (faster onset and shorter duratio than SC )
Insulin Delivery Systems
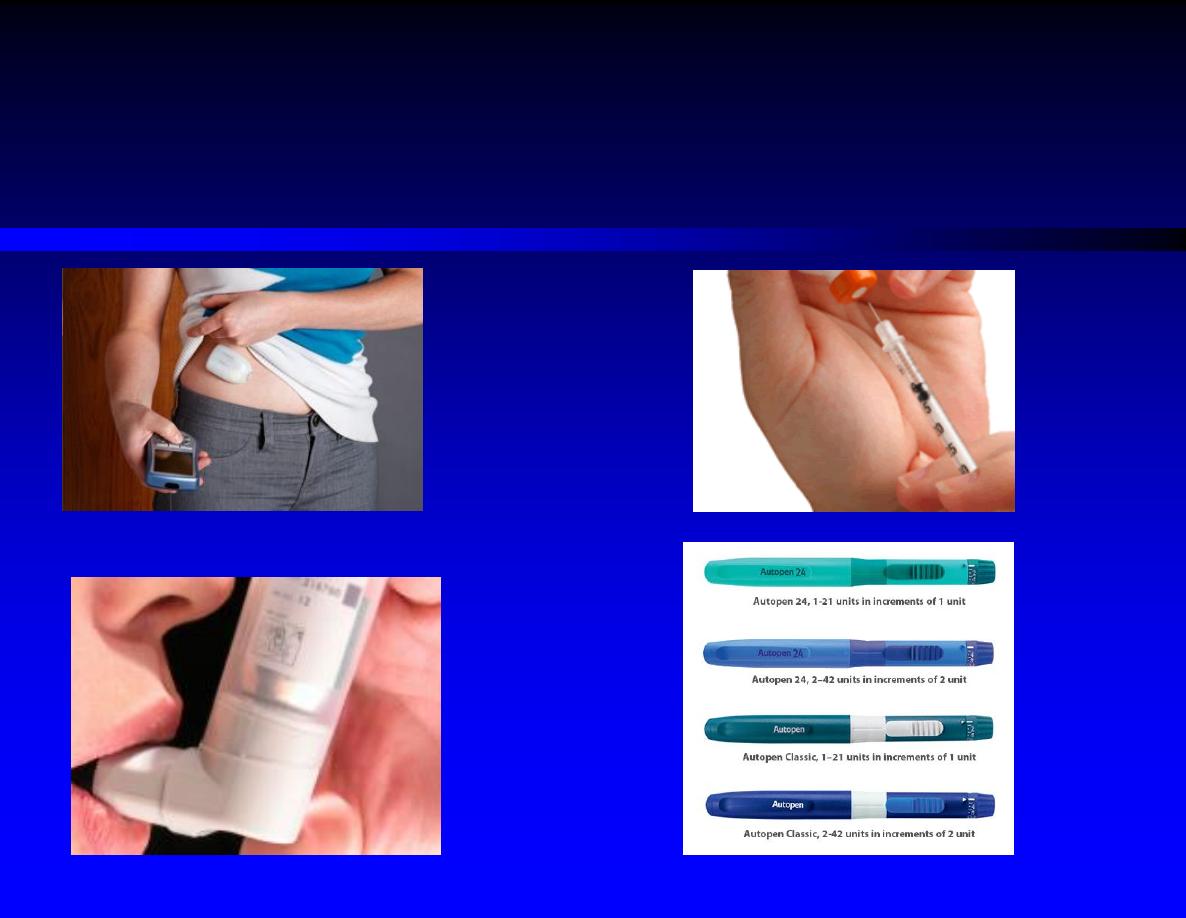
Insulin Delivery Systems

1) Hypoglycemia:
is the most common complication of insulin therapy. They usually result from
(Overdose of insulin, Excessive (unusual) physical exercise, meal is missed)
Mild Hypoglycemia and conscious patient treated by dextrose tab, glucose
gel or any sugar containing food or beverages
Severe hypoglycemia and disoriented or unconscious patient best treated by
IV 20-50 ml of 50% glucose over 2-3 minutes. Alternatively SC or IM 1mg
of glucagon. If patient still stuporus or glucagon not available, small amount
of honey or syrup can be inserted into buccal pouch.
Complications of Insulin Therapy

2)
Immunopathology of Insulin Therapy
:
Local or systemic allergic reactions
Immune insulin resistance, A low titer of circulating IgG anti-insulin
antibodies that neutralize the action of insulin to a negligible extent develops
in most insulin-treated patients.
3)
Lipodystrophy at Injection Sites
(with new prparation, atrophy less,
hypertrophy more)
4)
Weight gain
Complications of Insulin Therapy

Promising
Thank You
